Practice Parameter for the Use of Stimulant Medications in the Treatment of Children, Adolescents, and Adults
Abstract
This practice parameter describes treatment with stimulant medication. It uses an evidence-based medicine approach derived from a detailed literature review and expert consultation. Stimulant medications in clinical use include methylphenidate, dextroamphetamine, mixed-salts amphetamine, and pemoline. It carries FDA indications for treatment of attention-deficit/hyperactivity disorder and narcolepsy.
More than 60 years ago, serendipitous observation revealed that the drug dl-amphetamine reduces the disruptive symptoms of hyperkinetic children. Today, four stimulant medications are available for clinical use: methylphenidate (MPH), dextroamphetamine (DEX), mixed-salts amphetamine (AMP), and pemoline (PEM). They are the most widely prescribed psychotropic medications for children, primarily in the treatment of attention-deficit/hyperactivity disorder (ADHD). Long thought of as a childhood disorder, ADHD is now known to persist into adolescence and adulthood, and adults are increasingly being treated with stimulants for this condition. Stimulants are also indicated for the treatment of narcolepsy, based on controlled studies.
This practice parameter will (1) review the literature pertinent to the clinical use of stimulants; (2) describe indications and contraindications for stimulant treatment, with an emphasis on judicious use; (3) describe the initiation and dosing of the various stimulant agents; (4) describe the side effects encountered in stimulant treatment; (5) discuss long-term maintenance using stimulant agents; and (6) discuss the combination of stimulants and other psychotropic agents in the treatment of comorbid conditions.
Executive summary
The treatment of patients with stimulant medications requires the consideration of many factors that cannot be fully conveyed in the brief executive summary. The reader is encouraged to review the entire practice parameter. Each recommendation in the Executive Summary is identified as falling into one of the following categories of endorsement, indicated by an abbreviation in brackets following the statement. These categories indicate the degree of importance or certainty of each recommendation.
“Minimal Standards” [MS] are recommendations that are based on substantial empirical evidence (such as well-controlled, double-blind trials) or overwhelming clinical consensus. Minimal standards are expected to apply more than 95% of the time, i.e., in almost all cases. When the practitioner does not follow this standard in a particular case, the medical record should indicate the reason.
“Clinical Guidelines” [CG] are recommendations that are based on limited empirical evidence (such as open trials, case studies) and/or strong clinical consensus. Clinical guidelines apply approximately 75% of the time. These practices should always be considered by the clinician, but there are exceptions to their application.
“Options” [OP] are practices that are acceptable but not required. There may be insufficient empirical evidence to support recommending these practices as minimal standards or clinical guidelines. In some cases they may be appropriate, but in other cases they should be avoided. If possible, the practice parameter will explain the pros and cons of these options.
“Not Endorsed” [NE] refers to practices that are known to be ineffective or contraindicated.
Brief history
Stimulants are among the most effective psychotropic medications in clinical use today. Their effects on disruptive behavior were discovered in 1937, when these drugs proved to increase compliance, improve academic performance, and reduce motor activity in hyperkinetic children. Studies of the short-term benefits of stimulants on the symptoms of ADHD constitute the largest body of treatment literature on any childhood-onset psychiatric disorder. By 1996, 161 randomized controlled trials had been published, encompassing 5 preschool, 150 school-age, 7 adolescent, and 9 adult studies. Improvement occurred in 65% to 75% of the 5,899 patients randomized to stimulants versus only 5% to 30% of those assigned to placebo for MPH (n=133 trials), DEX (n=22 trials), and PEM (n=6 trials). Over the past two decades, there has been a steady increase in the diagnosis of ADHD and the use of stimulants, particularly in the United States. Because stimulant medications can be abused, the rapid increase in stimulant use has raised concerns about the risks of diversion and abuse. In part because of these concerns, the use of stimulants to treat children remains controversial, particularly in the lay media and Internet. As always, practitioners should exercise care in making an accurate diagnosis.
Psychopharmacological effects of stimulants
Short-term trials have reported improvements in the most salient and impairing behavioral symptoms of ADHD. Except for PEM, the immediate release preparations of the major stimulants have a brief duration of action, providing clinical benefits for 3 to 5 hours after oral dosing. This requires multiple doses during the day to maintain improvement. In the classroom, stimulants decrease interrupting, fidgeting, and finger tapping and increase on-task behavior. At home, stimulants improve parent-child interactions, on-task behaviors, and compliance. In social settings, stimulants improve peer nomination rankings of social standing and increase attention during sports activities. Stimulants decrease response variability and impulsive responding on laboratory cognitive tasks, increase the accuracy of performance, and improve short-term memory, reaction time, math computation, problem-solving in games, and sustained attention. Time-response studies show a differential impact across symptom domains, with behavior affected more than attention. Stimulants continue to ameliorate the symptoms of ADHD in the presence of other comorbid Axis I disorders and may even show positive benefit on the comorbid disorder (such as conduct disorder and anxiety disorder).
Until recently, the benefits of stimulant treatment have been demonstrated only in short-duration trials, most lasting less than 12 weeks. To address this issue, prospective, longer-duration randomized controlled trials lasting 12 to 24 months have been conducted. Doses up to 50 mg/day of MPH were used in these long-duration studies. The largest of these studies, the NIMH Collaborative Multisite Multimodal Treatment Study of Children With Attention-Deficit/Hyperactivity Disorder (MTA study), showed that stimulants (either by themselves or in combination with behavioral treatments) lead to stable improvements in ADHD symptoms as long as the drug continues to be taken.
Although there are only a few randomized controlled trials documenting their efficacy, stimulants have proved effective in the treatment of narcolepsy.
Indications
A clinician determines that a patient (child, adolescent, or adult) has a condition indicated for the use of stimulant medications [MS].
Psychiatric evaluation should include a detailed history (psychiatric and medical) of the patient, collateral information from parents or significant others, documentation of target symptoms, and a mental status examination. It is helpful to gather information from at least two adult sources—preferably from different settings in a child’s life (e.g., home or school)—about the child’s symptoms. Conditions that may be the focus of stimulant use are:
| • | ADHD. The clinician should document that the patient has the DSM-IV or ICD-10 diagnosis of ADHD. There is no empirically proven threshold of ADHD symptoms that can be used to predict treatment response to stimulant medication. Fortunately, the ratio of benefit to side effects is very favorable for MPH, DEX, and AMP. The severity of the symptoms and the resulting impairment in the patient’s academic or occupational, social, and family functioning should be assessed. Only those patients with moderate to severe impairment in two different settings should be considered for stimulant treatment. A child with ADHD, predominantly inattentive type with severe academic problems at school and during homework, may be considered for stimulant treatment, even if his peer relationships and family functioning are not otherwise affected. Teacher ratings of ADHD symptoms, using a validated and age- and sex-normed instrument, should be obtained at baseline and after treatment with stimulants [CG]. To qualify for treatment, the child should be living with a responsible adult who can administer the medication; the school should also provide personnel for supervising in-school doses. In addition to stimulants, consider other effective modalities, such as parent training, psychoeducation, and others, as described in the Academy’s Practice Parameters for ADHD (American Academy of Child and Adolescent Psychiatry, 1997a). | ||||
| • | ADHD comorbid with conduct disorder. Only those patients with symptoms that cause moderate to severe impairment in at least two different settings should be considered for stimulant treatment. If the child is an adolescent, the clinician should be certain that the patient is not using nonprescribed stimulants [CG]. | ||||
| • | Narcolepsy. The patient suffers from excessive sleepiness with recurrent sleep attacks and cataplexy (brief episodes of bilateral weakness typical of the rapid eye movement phase of sleep, even though the individual is awake) [CG]. | ||||
| • | Apathy due to a general medical condition. Individuals who have suffered a brain injury due to a cerebrovascular accident, trauma, HIV, or a degenerative neurological illness often exhibit apathy or symptoms of inattention and impulsivity similar to ADHD. If the illness or trauma occurred after age 7, they would not meet criteria for ADHD. Clinical experience and small controlled trials suggest that stimulants are helpful in reducing such behaviors in these patients [OP]. Doses of the stimulants are typically lower than those used in the treatment of ADHD. | ||||
| • | Adjuvant medical uses of stimulants. Some severely medically ill patients develop severe psychomotor retardation secondary to the illness itself, the sedative effects of pain medication, or toxic effects of the agents used to treat the primary illness (i.e., chemotherapy for cancer). Case reports suggest that low doses of stimulants may enable these patients to be more alert and have a higher energy level and better appetite [OP]. | ||||
| • | Treatment-refractory depression. Stimulants, particularly MPH, have been used to augment the effects of tricyclic antidepressants [OP]. Doses are usually lower than used to treat ADHD. | ||||
Contraindications
Contraindications to the use of stimulants in clinical practice include previous sensitivity to stimulant medications, glaucoma, symptomatic cardiovascular disease, hyperthyroidism, and hypertension. These medications must be used with great care if there is a history of drug abuse. They are contraindicated in patients with a history of illicit use or abuse of stimulants, unless the patient is being treated in a controlled setting or can be supervised closely [NE]. If a member of the household has a history of use or abuse of stimulants, steps should be taken to make certain that the medications prescribed are not abused. Concomitant use of a monoamine oxidase (MAO) inhibitor is contraindicated [NE]. Stimulants should not be administered to a patient with an active psychotic disorder [NE].
The Food and Drug Administration–approved package inserts add other contraindications, including motor tics, marked anxiety, and a family history or diagnosis of Tourette’s disorder. The recent clinical trial literature, however, reveals that these conditions may not be worsened by stimulant treatment. Because the package insert mentions that MPH lowers the seizure threshold, it is best to initiate MPH after the seizure disorder is under control with anticonvulsants. There are published studies showing that epileptic patients taking anticonvulsants do not show a change in seizure frequency when MPH is added. The package insert warns against starting MPH in children under the age of 6 years, although there are now 8 published reports finding that MPH is effective in this age range. On the other hand, the package inserts for PEM, DEX, and mixed salts of AMP allow their use in children down to age 3 years, even though there are no published controlled studies of these drugs in preschoolers.
Use of stimulants
Using stimulant medication in treating patients with ADHD or ADHD plus conduct disorder requires careful documentation of prior treatments, selection of the order of stimulants to be used, using the recommended starting dose of each stimulant, deciding on both a minimum and maximum dose, using a consistent titration schedule, deciding on a method of assessing drug response, managing treatment-related side effects, and providing a schedule for the monitoring of long-term medication maintenance [CG].
| • | Documentation of prior treatment. Documen-tation of adequate assessment, previous psychosocial treatments, and previous psychotropic medication treatments should be done before stimulant treatment is initiated [MS]. Information collected should include the name of the medication, dosage, duration of the trial, response and side effects, and estimation of compliance. Other useful information may include special school placements and psychosocial treatments including behavioral modification, parent training, and daily report card. | ||||
| • | Obtaining a baseline blood pressure, pulse, height, and weight in the context of a physical examination. All children should have a routine physical examination before starting stimulant medications. This physical should include vital signs, including blood pressure, pulse, height, and weight. This will help discover adolescents and younger children who may have malignant hypertension and adults who have essential hypertension and/or cardiac arrhythmias. Children should have their vital signs checked annually during their routine physical examination. Adults on stimulants should have blood pressure and pulse checked on a quarterly basis by the treating physician or by the primary care physician. | ||||
| • | Selecting the order of stimulants to be used. The first stimulant used may be MPH, AMP, or DEX, depending on clinician and patient preference. On average, however, the problematic effects on appetite and sleep are greater with AMP or DEX, consistent with their longer excretion half-lives. PEM is not recommended by this parameter because, although it is effective, it may lead to hepatic failure. | ||||
| • | Using the recommended starting dose of each stimulant. The starting doses of stimulants are 5 mg for MPH and 2.5 mg for DEX/AMP, generally given in the morning after breakfast and around noon after lunch. | ||||
| • | Deciding on both a minimum and maximum dose. For children and adolescents, minimum effective doses should be used to initiate therapy. A minimum starting dose is either 5 mg of MPH or 2.5 mg of AMP in children and adolescents, given in the form of an immediate-release tablet. These doses should be started on a 2 or 3 times daily basis because of their very short duration of action. The maximum total daily doses are calculated by adding together all doses taken during a given day. The Physician’s Desk Reference (PDR) states that the maximum total daily dose is 60 mg for MPH and 40 mg for amphetamines. Children weighing less than 25 kg generally should not receive single doses greater than 15 mg of MPH or 10 mg of DEX/AMP. The consensus from practice is that doses may go higher than the PDR-recommended upper limits on rare occasions. Experts often limit the upper range to a total daily dose of 40 mg of AMP or 25 mg for a single dose of MPH, when MPH is given in multiple doses throughout the day. If the top recommended dose does not help, more is not necessarily better. A change in drug or environmental or psychosocial intervention may be required. | ||||
| • | Using a consistent titration schedule. If symptom control is not achieved, the dose generally should be increased in weekly increments of 5 to 10 mg per dose for MPH or 2.5 to 5 mg for DEX/AMP [CG]. Alternatively, the physician may elect to use a fixed-dose titration trial, similar to that found in the MTA study, in which a full set of different doses is switched on a weekly basis. At the end of such a trial, the parent and physician can meet to decide which dose worked best for the child. The advantage for such a full dose trial is that a child is less likely to miss a high dose that might yield additional improvement [OP]. | ||||
| • | Deciding on a method of assessing drug response. Follow-up assessment should include evaluation of target symptoms of ADHD, asked regularly of the parent and of a teacher [CG]. These clinical assessments may be supplemented by the use of parent and teacher rating scales. It is important to obtain self-ratings from adolescents and from adults. | ||||
| • | Managing treatment-related side effects. Side effects should be systematically assessed by asking specific questions of patients and of parents about known side effects, such as insomnia, anorexia, headaches, social withdrawal, tics, and weight loss [CG]. Weighing the patient at each visit provides an objective measure of loss of appetite. | ||||
| • | Providing a schedule for initial titration and monitoring [CG]. During initial titration and during later drug dose adjustments, contact can be maintained on a weekly basis by telephone [CG]. The titration phase of stimulant initiation covers the period of dose adjustment and often requires 2 to 4 weeks. | ||||
| • | Providing a schedule for monitoring the drug maintenance phase: Afterward, patients can be followed up regularly for lengthy periods on the same dose, and are said to be in a maintenance phase. Follow-up appointments should be made at least monthly until the patient’s symptoms have been stabilized [MS]. Changes in the frequency of physician visits should be governed by robustness of drug response, adherence of the family and patient to a drug regimen, concern about side effects, and need for psychoeducation and/or psychosocial intervention. More frequent appointments should be made if there are side effects, significant impairment from comorbid psychiatric disorders, or problems in adherence to taking the stimulants. The response and severity of the patient’s symptoms determine the frequency of appointments. Optional treatment components include the collection of teacher reports before or at each visit, provision of reading materials, and discontinuation trials. | ||||
Complications and side effects
Almost all stimulant-related side effects reported for children and adolescents with ADHD are rare and short-lived and are responsive to dose or timing adjustments. Mild side effects are common, and serious side effects are rare and short-lived if the medication is reduced in dose or discontinued. Severe movement disorders, obsessive-compulsive ruminations, or psychotic symptoms are very rare and disappear when the medication is stopped. It was recently determined that patients on PEM experience hepatic failure 17 times more frequently than the spontaneous rate; this rare but serious side effect is a major complication of PEM usage. In placebo-controlled studies of stimulants, parents report only seven side effects occurring more often on stimulant than on placebo: delay of sleep onset, reduced appetite, weight loss, tics, stomach-ache, headache, and jitteriness. Careful lowering of the dose or changing of the timing of the dose administration may alleviate the side effect [CG]. When insomnia or appetite loss occurs but the stimulant is highly beneficial in reducing the target symptoms, a variety of adjunctive tactics are available to ameliorate the side effects. Staring, daydreaming, irritability, anxiety, and nailbiting may typically decrease with increasing dose, representing preexisting symptoms rather than side effects.
Literature review
The literature on stimulant treatment of children with attention-deficit/hyperactivity disorder (ADHD) is voluminous. Books and journals published from 1980 through the end of 2000 were reviewed in detail; older references were included when pertinent. A National Library of Medicine search using the keywords dextroamphetamine, methylphenidate, pemoline, and Adderall® ensured completeness of coverage. Using Freedom of Information Letters, the Food and Drug Administration supplied data on spontaneous postmarketing reports of side effects from psychostimulants. In addition, the authors drew on their own experience.
Brief history
The behavioral effects of stimulants were discovered more than 60 years ago (Bradley, 1937). dl-Amphetamine, the racemic form of amphetamine (AMP), produced a dramatic calming effect while simultaneously increasing compliance and academic performance. Over the next two decades, Bradley published case reports of children improving during AMP treatment (Bradley and Bowen, 1941). Subsequent studies showed that psychostimulants (AMP only) increased the seizure threshold (Laufer et al., 1957), decreased oppositional behavior of boys with conduct disorder in a residential school (Eisenberg et al., 1961), and reliably improved the target symptoms of ADHD on standardized rating forms filled out by parents and teachers (Conners et al., 1967).
In the years following, many short-term controlled treatment studies revealed that psychostimulants were effective, with most protocols lasting between 1 and 3 months. Between 1962 and 1993 there were more than 250 reviews and more than 3000 articles on stimulant effects (Swanson, 1993). Reviews of controlled studies (American Academy of Child and Adolescent Psychiatry, 1997a; Barkley, 1977, 1982; DuPaul and Barkley, 1990; Gittelman-Klein, 1980, 1987; Schmidt et al., 1984), have demonstrated beneficial stimulant effects for children with ADHD during brief trials.
Stimulant prescribing in the United States
Data from diverse sources suggest a steeply rising rate of stimulant prescribing in the United States during the past decade. ADHD-related outpatient visits to primary practitioners increased from 1.6 to 4.2 million per year during the years 1990 to 1993 (Swanson et al., 1995). During those visits, 90% of the children were given prescriptions, 71% of which were for the stimulant methylphenidate (MPH). During the same period, MPH production in the United States increased from 1,784 kg/yr to 5,110 kg/yr. More than 10 million prescriptions for MPH were written in 1996 (Vitiello and Jensen, 1997). Recent epidemiological surveys have estimated that 12-month stimulant prescription rates range from 6% in urban Baltimore (Safer et al., 1996) to 7.3% in rural North Carolina (Angold et al., 2000). One epidemiological survey found that up to 20% of white boys in fifth grade in one location were receiving medication for ADHD (LeFever et al., 1999).
Experts have speculated that increased MPH production quotas and prescriptions written could be due to improved recognition of ADHD by physicians, an increase in the prevalence of ADHD (Goldman et al., 1998), or an easing of the standards for making the ADHD diagnosis or a relaxation of the standards for dispensing stimulants. The increase has been attributed to lengthened duration of treatment and the inclusion of children with learning disabilities, more adolescents, more girls, children with ADHD-Inattentive Type, and adults with ADHD (Safer et al., 1996). A 1998 Consensus Development Conference on ADHD sponsored by the National Institutes of Health (NIH Consensus Statement, 1998) found “wide variations in the use of psychostimulants across communities and physicians.”
This wide variability in practice was attributed to a lack of a well-understood, universally-accepted “ADHD diagnostic threshold above which the benefits of psycho-stimulant therapy outweigh the risks.” Are clinicians “catching up” in their appreciation of how many children have true ADHD or overprescribing?
Epidemiological surveys that include child diagnoses and treatment services have given divergent answers to this question. One survey in 4 different communities found that only one-eighth of the children who met criteria for ADHD received adequate stimulant treatment (Jensen et al., 1999), while another survey in rural North Carolina found that 72% of school-age children on stimulants did not meet criteria for ADHD (Angold et al., 2000).
The U.S. Drug Enforcement Administration has been concerned about the risk of abuse and diversion of these medications, particularly when the media report that college students grind up immediate-release stimulant tablets and snort the powder. Analyses of annual school surveys of drug use and the Drug Abuse Warning network data on emergency room visits, however, have not suggested increased abuse or diversion of MPH.
Psychopharmacology
Although the psychostimulants are the medications of choice for the treatment of children with ADHD, their central mechanisms of action are unknown. Studies using positron emission tomography (PET) scanning have demonstrated that untreated adults with a past and current history of ADHD showed 8.1% lower levels of cerebral glucose metabolism than controls (Zametkin et al., 1991), with the greatest differences in the superior prefrontal cortex and premotor areas. MPH and dextroamphetamine (DEX) elevate glucose metabolism in the brains of rats, although patients with schizophrenia given DEX show decreased glucose metabolism. No consistent changes in cerebral glucose metabolism were found in PET scans done before and on medication in 19 MPH-treated and 18 DEX-treated adults with ADHD, even though the adults showed significant improvements in behavior (Matochik et al., 1993).
Various theories of the pathophysiology of ADHD have evolved, most depicting problems in brain frontal lobe function. Recent theories of dysfunction in ADHD focus on the prefrontal cortex, which controls many executive functions (e.g., planning, impulse control) that are impaired in ADHD. Stimulants used for treatment of children with ADHD have putative effects on central dopamine and norepinephrine pathways that are crucial in frontal lobe function. Stimulants act in the striatum by binding to the dopamine transporter, with a resulting increase in synaptic dopamine. This may enhance the functioning of executive control processes in the prefrontal cortex, ameliorating the deficits in inhibitory control and working memory reported in children with ADHD (Barkley, 1997).
PET scans of adult volunteers have added useful data. When given orally, [11C]MPH occupies a high proportion of dopamine transporter sites in the striatum but is not associated with euphoria, which is found after intravenous administration (Swanson and Volkow, 2000; Volkow et al., 1998). Acute administration of stimulant medications increases norepinephrine and dopamine in the synaptic cleft, but whether compensatory mechanisms occur after slower oral absorption is not known (Grace, 2000).
The pharmacokinetics of the stimulants are characterized by rapid absorption, low plasma protein binding, and rapid extracellular metabolism (Patrick et al., 1987). Although several pathways, including p-hydroxylation, N-demethylation, deamination, and conjugation are involved in their metabolism, up to 80% may be excreted unchanged in the urine, in the case of AMP, or undergo de-esterification in plasma, as in the case of MPH (Patrick et al., 1987). Multiple doses are necessary to sustain behavioral improvements during school, recreational activities, and homework. Both absorption and bioavailability may increase after a meal (Chan et al., 1983). When the dose is weight-adjusted, there are no age effects on dose. Generic MPH and the brand name product show similar but not identical pharmacokinetic profiles. The generic is absorbed more quickly and peaks sooner (Vitiello and Burke, 1998).
Stimulants are rapidly absorbed from the gut and act within the first 30 minutes after ingestion. Effects on behavior appear during absorption, beginning 30 minutes after ingestion and lasting 3 to 4 hours. Plasma half-life ranges between 3 hours (for MPH) and 11 hours (for DEX). The concentration-enhancing and activity-reducing effects of MPH can disappear well before the medication leaves the plasma, a phenomenon called “clockwise hysteresis” (Cox, 1990).
Stimulant medication effects on ADHD are concentrated within the early part of the absorption phase (Perel et al., 1991). The rate of absorption of psychostimulants is very rapid, delivering a quick, large peak in plasma concentration. Monoamine neurotransmitters pulse into the synaptic cleft during this rapid stimulant concentration change. This bolus was thought to be necessary for the stimulant-related reduction in ADHD symptoms, so that drugs that were absorbed rapidly produced more improvement than stimulants with a gradual increase (e.g., sustained-release MPH). A steep slope of stimulant medication absorption from immediate-release stimulants was thought to be necessary for producing robust improvement and was called the “ramp effect” (Birmaher et al., 1989). More recent studies, however, have shown that a gradual ascending increase in MPH plasma concentration over the day—without a bolus or sharp ramp-up in absorption—produces the equivalent reduction in ADHD symptoms to the three peaks from immediate-release MPH in a 3-times-daily schedule (Swanson et al., 1999a). This became the basis for the design of the OROS-MPH release pattern.
Pemoline (PEM) effects on cognitive processing, like MPH, begin within the first 2 hours after administration (Sallee et al., 1992). Unlike MPH, the effects last up to 6 hours. While the therapeutic effects of MPH and DEX are confined to the absorption phase, PEM has a significant postabsorptive effect lasting into the postdistribution phase. Unlike previous clinical suggestions that PEM requires 3 to 6 weeks to work (Page et al., 1974), PEM has been shown to be effective after the first dose (Pelham et al., 1995; Sallee et al., 1985).
Overall, there has been little evidence of the development of tolerance to the stimulant effects on symptoms of ADHD and little evidence of a need to increase the dose to get the same response (Safer and Allen, 1989). Children most often continue to respond to the same dose of stimulant medication, even though early studies had suggested that upward dose adjustment might be required after several months of treatment (Satterfield et al., 1979). More recent pharmacodynamic studies suggest that stimulant blood levels need to increase throughout the day to maintain constant efficacy. This is because short-term tolerance to MPH develops by the second dose given in the same day (Swanson et al., 1999a).
Another concern has been raised regarding long-term treatment with stimulants. An uncontrolled follow-up study suggested that long-term treatment with MPH might predispose children with ADHD to abuse of nicotine and possibly also cocaine (Lambert and Hartsough, 1998). This speculation was based on the process of sensitization, a progressive increase in a drug effect with repeated treatment. This has been shown to influence two types of animal behavior, locomotion/stereotypy and incentive motivation (Robinson and Camp, 1987; Shuster et al., 1982).
Sensitization in animals is facilitated by high doses of MPH (relative to clinical doses), different routes of administration (intravenous or intraperitoneal rather than oral), and a different schedule of administration (intermittent rather than chronic) than used in the treatment of children with ADHD. This makes it difficult to extrapolate from animal findings to the use of MPH to treat children with ADHD.
Classic stimulant effects in adults include a prolongation of performance at repetitive tasks before the onset of fatigue, a decreased sense of fatigue, mood elevation, euphoria, and increased speech rate (Rapoport et al., 1980). The psychostimulants increase CNS alertness on tasks requiring vigilance, both in laboratory tasks, such as the Continuous Performance Task (CPT), or on the job, such as maintaining the ability to notice new events on a radar screen over periods of hours. Stimulants decrease response variability and impulsive responding on cognitive tasks (Tannock et al., 1995b); increase the accuracy of performance; and improve short-term memory, reaction time, seatwork computation, problem-solving in games with peers (Hinshaw et al., 1989), and sustained attention.
Children and adolescents respond similarly to stimulants. In the classroom, stimulants decrease interrupting, fidgeting, and finger tapping and increase on-task behavior (Abikoff and Gittelman, 1985). At home, stimulants improve parent-child interactions, on-task behaviors, and compliance; in social settings, stimulants improve peer nomination rankings of social standing and increase attention while playing baseball (Richters et al., 1995).
Stimulant drugs have been shown to affect children’s behavior cross-situationally (classroom, lunchroom, playground, and home) when these drugs are administered repeatedly throughout the day. Time-response studies of stimulant effects, however, show a different pattern of improvement for behavioral and for attentional symptoms, with behavior affected more than attention. For example, a controlled, analog classroom trial (n=30) of AMP (Swanson et al., 1998) revealed rapid improvements in teacher ratings of behavior, while changes in math performance occurred later, about 1.5 hours after administration. The duration of improvement was dependent on dose.
The pharmacodynamic effects on behavior of the immediaterelease formulations of MPH and DEX appear within 30 minutes, reach a peak within 1 to 3 hours, and are gone by 4 to 6 hours (Swanson et al., 1978, 1998). This “roller-coaster effect,” plus missed doses and irregular compliance, all complicate the treatment picture. In-school dosing is a necessity for most children on immediate-release stimulants. This requires additional supervision by school personnel and increases the risk of peer ridicule. Clinicians and parents report that some children have intense wear-off effects (“rebound”) in the late afternoon. Controlled studies using actometers and analog classrooms—perhaps not ecologically relevant to the real environment of an ADHD child—have not been able to confirm these reports.
When medication is discontinued, its effects cease. One double-blind discontinuation study using DEX (Gillberg et al., 1997), however, found that the ADHD-symptom reduction from 15 months of treatment with DEX continued after the drug was stopped. A small proportion of children with ADHD have been reported to respond sufficiently to single-day dosing with immediate-release MPH (Pliszka, 2000). Sustained-release formations of MPH and DEX, as well as PEM, have been shown to have effects on laboratory tests of vigilance for up to 9 hours after dosing (Pelham et al., 1990a). Clinicians have found, however, that these drugs may not successfully cover the entire school day with only one morning dose.
Long-acting stimulants
The need for long-duration drugs emanates from a variety of concerns. The time-response characteristics of standard stimulants are such that the plasma level troughs occur at the most unstructured times of the day, such as lunchtime, recess, or during the bus ride home from school (Pelham et al., 2000). Compliance is also a problem with standard, short-duration stimulants. Schools may not reliably administer the medication or may have policies that prohibit its administration. Some children—especially adolescents—avoid cooperating with in-school dosing because of fear of ridicule and a wish for privacy. Other children with ADHD simply forget to take their afternoon doses.
Stimulant-induced reduction of impulsivity improves peer interactions during recreational activities (Pelham et al., 1990b; Pelham and Waschbusch, 1999). When the stimulants are given after school, children may be in day care, be playing sports, or be riding a school bus at the time the dose should be administered. The conflict of a child’s daily schedule with the tight time demands of short-acting stimulants often interferes with adherence to treatment schedules and with obtaining the best clinical results from the medication.
Long-duration versions of the stimulant medications have been available for more than a decade. Ritalin-SR® (MPH-SR20) uses a wax-matrix vehicle for slow release, whereas the DEX Spansule® is a capsule containing small medication particles. There are slow-release generics (Methylphenidate-SR, Metadate®, Methylin-SR®) that use the same basic wax-matrix mechanism for sustained release as found in Ritalin-SR. Yet use by clinicians has been far less than expected.
Clinicians find the long-duration MPH less effective than the short-acting version. Pelham and colleagues first reported that MPH-SR20 was less effective, according to a panel of expert raters who reviewed behavioral and CPT data, than the standard MPH 10 mg bid, when both were used to treat 13 children with ADHD in a summer program (Pelham et al., 1989). In a later study (Pelham et al., 1990a), the same investigators reported that MPH-SR20 was equally effective as DEX Spansules or PEM for maintaining attention on a task over a 9-hour period.
MPH immediate-release (MPH-IR) produces higher peak plasma concentrations and yields a steeper absorption-phase slope (“ramp” effect) than does the longer-acting MPH-SR20 preparation, as shown in a study of nine males with ADHD in which equal doses of MPH were delivered by an MPH-IR20 mg tablet or as an MPH-SR20 tablet (Birmaher et al., 1989). Because MPH-SR20 is designed to release more slowly, a comparison for matching peak effects between a 10-mg MPH-IR and a 20-mg MPH-SR20 tablets would be more equitable. MPH-SR20 begins to act 90 minutes after ingestion (compared with 30 minutes for the MPH-IR preparation), and its plasma level peak is lower than for a comparable dose of MPH-IR. Behavioral and cognitive studies show that the peak benefit for the MPH-SR20 occurs at 3 hours, 1 hour later than for the standard preparation (Pelham et al., 1989).
Sustained-release MPH may not be as immediately helpful to children with ADHD, for several reasons. MPH-SR has a delayed onset of action and a gradually decreasing plasma concentration after its peak at 3 hours (Birmaher et al., 1989). A recent pharmacodynamic study in laboratory classroom settings revealed that if the children’s afternoon doses are identical to or smaller than those in the morning, their ADHD symptoms increase (Swanson et al., 1999a). Although another laboratory classroom study (Pelham et al., 2000) failed to replicate this effect, afternoon attenuation could explain the lower efficacy of MPH-SR-20.
New long-acting stimulants
Pediatric psychopharmacological drug development by the pharmaceutical industry has increased greatly in the past 3 years. Most new drugs are targeted for children with ADHD. A number of the “new” treatments for ADHD address the need for a more effective single-dose-per-day, long-duration stimulant. Children with ADHD now on immediate-release stimulants or the older variety long-duration preparations, such as MPH-SR20 or Dexedrine Spansules, can be switched to these newer preparations. The new medication called OROS-MPH (Concerta®) has been shown to be a useful alternative to older stimulant medications (Swanson et al., 2000) in a community-based study. OROS-MPH given once daily in the morning was shown to be equally effective as MPH immediate release tablets given 3 times daily in a double-blind, placebo-controlled, 14-site, randomized controlled trial (Wolraich et al., in press). Concerta given once a day produces an ascending-pattern plasma drug level generated by the caplet’s osmotically released, timed drug-delivery system. Children naïve to stimulant treatment may be started directly on the 18-mg Concerta, which is equivalent to MPH 5 mg 3 times daily.
Plasma levels of stimulants
MPH plasma levels do not correlate with clinical response (Gualtieri et al., 1982) and provide no more predictive power than teacher and parent global rating forms (Sebrechts et al., 1986).
Toxicology
Animal toxicity studies using high doses of stimulants have reported abnormal findings not found in humans. This may be a result of differences of species, dose, route of administration, and end point selected. Sprague-Dawley rats given high-dose (25 mg/kg subcutaneous versus 0.3 mg/kg orally in children) injections of DEX, MPH, methamphetamine, and 3,4-methylene-dioxymethamphetamine have shown loss of serotonin reuptake sites (Battaglia et al., 1987). Hepatic tumors increased only in mice (a strain known to have genetic diathesis for liver tumors), whereas rats had a decreased rate (similar to human data) when treated with high, 4- to 47-mg/kg oral doses of MPH (Dunnick and Hailey, 1995). The 1998 NIH Consensus Development Conference on ADHD cautioned that extremely high doses of stimulants might cause central nervous system damage, cardiovascular damage, and hypertension (NIH Consensus Statement, 1998). Single doses 50 times that used in children have produced such severe effects, as found in Japanese factory workers who took large amounts of amphetamines to work long hours in postwar Japan. These effects relate far more to conditions of severe toxic overdose than to standard practice. Paranoid hallucinations have been produced in normal adult human volunteers by single doses of 300 mg of AMP (Angrist and Gershon, 1972).
Effectiveness of stimulant medications
Therapeutic effects of stimulants
Short-term trials of stimulants, most often 3 months or less in duration, have reported robust efficacy of MPH, DEX, and PEM, with equal efficacy among stimulants (McMaster University Evidence-Based Practice Center, 1998). More than 160 controlled studies involving more than 5,000 school-age children—only 22 lasting more than 3 months (Schachar and Tannock, 1993)—demonstrated a 70% response rate when a single stimulant is tried (Spencer et al., 1996b). Short-term trials have reported improvements in the most salient and impairing behavioral symptoms of ADHD, including overt aggression, as long as medication is taken. Individual children show different responses and improvements, with fewer than half of the children showing normalization. Therefore, children with ADHD taking stimulant medication continue to have more behavior problems than those with no history of mental disorder. Although many recent studies have shown distinct improvements in daily academic performance, there have been no long-term, controlled prospective studies of the academic achievement and social skills of children with ADHD treated with stimulants in a consistent manner.
Stimulant treatment leads to improvements in both ADHD symptoms and associated conditions compared with placebo, other drug classes, or nonpharmacological treatments (Greenhill, 1998a; Jacobvitz et al., 1990; Spencer et al., 1996b; Swanson, 1993). Effect sizes for changes in behavior or attention in short-term trials range from 0.8 to 1.0 SD on teacher reports (Elia et al., 1991; Thurber and Walker, 1983) for both MPH and DEX. Stimulant medications improve behavior and attention in children with other disorders and in normal subjects, so these drug effects on behavior are neither “paradoxical” nor specific for ADHD (Rapoport et al., 1980). Therefore, a positive response to stimulants is not diagnostic for ADHD. Stimulant medications have been reported to be helpful in other medical conditions, such as narcolepsy and depression (Goldman et al., 1998).
Stimulant effects on comorbid psychiatric disorders
Two-thirds of children with ADHD present with one or more comorbid Axis I psychiatric disorders, primarily oppositional defiant disorder, conduct disorder, or anxiety disorder (MTA Cooperative Group, 1999a). Comorbid symptoms may alter the response to stimulants. Children with ADHD and comorbid anxiety disorders initially were reported to have shown increased placebo response rates (DuPaul et al., 1994; Pliszka, 1992), a greater incidence of side effects, and smaller improvements on cognitive tests (Tannock et al., 1995a) while being treated with MPH. More recent controlled studies have shown no moderating effects of comorbid anxiety on treatment outcome when children with ADHD are treated with MPH (Diamond et al., 1999; MTA Cooperative Group, 1999b). Controlled studies of children with both Tourette’s disorder and ADHD have shown a variable impact on tic frequency patterns (Castellanos et al., 1997; Gadow et al., 1995). In a controlled study of 84 boys with ADHD and comorbid conduct disorder, ratings of antisocial behavior specific to conduct disorder were significantly reduced by MPH treatment even when one subtracts out the effect of improvement of the stimulants on the child’s ADHD baseline symptoms (Klein et al., 1997). Stimulants do not precipitate young adult bipolar disorders in boys comorbid for both ADHD and nonpsychotic bipolar disorder on mood stabilizers, either acutely or later on (Carlson et al., 2000).
Long-term trials of stimulant medications
Clinicians are interested in whether stimulant medications will continue to ameliorate the symptoms of ADHD when used long-term, as they do in practice. Uncontrolled, open, longer-duration retrospective studies published in the late 1970s reported that stimulant-treated children did not maintain their initial social or academic improvements. These longer-term reports, however, were flawed by retrospective methods, nonrandom assignment, nonstandard outcome measures, irregular stimulant prescribing patterns (Sherman, 1991), and the failure to include measures of adherence to the medication regimen (Schachar and Tannock, 1993). Even if these methodological problems were to be addressed, it would be ethically impossible to run multiyear controlled studies of stimulants, because of the requirement for maintaining large numbers of children with ADHD on placebo or ineffective control treatments for years, when treatments of proven short-term efficacy are available.
Prospective, longer-duration stimulant treatment trials use innovative control conditions, such as community standard care (Arnold et al., 1997), double-blind placebo discontinuation (Gillberg et al., 1997), or putting all children on stimulants and then comparing additional treatments (Abikoff and Hechtman, unpublished, 1998). These studies have shown maintenance of stimulant medication effects over periods ranging from 12 months (Gillberg et al., 1997) to 24 months (Abikoff and Hechtman, unpublished, 1998).
The MTA study compared treatment with stimulants alone, stimulants used in combination with intensive behavioral therapy (multimodal therapy), intensive behavioral therapy alone, and treatment as usual in the community for 579 children with ADHD, ages 7 to 9 years, treated over a 14-month period. Details of the MTA medication treatment protocol, which uses a strategy to enhance treatment response, have been published elsewhere (Greenhill et al., 1996). The results showed that optimally titrated MPH was more effective than intensive behavioral therapy, that combined treatment was more effective than behavioral treatment, and that all three MTA treatments were better than routine care in the community (MTA Cooperative Group, 1999a). Baseline characteristics, such as patient’s gender or presence of an anxiety disorder, did not affect the response to stimulant medications. That confirms previous reports that girls and boys respond equally well to stimulant medications.
In addition to the MTA study, there have been three other stimulant medication randomized controlled trials that have lasted 12 months or longer (Abikoff and Hechtman, unpublished, 1998; Gillberg et al., 1997; Schachar et al., 1997). The Gillberg study examined children comorbid for ADHD and pervasive developmental disorder and showed good response to DEX, although the small number in the study (n=62) prevented conclusive proof that pervasive developmental disorder does not affect response to stimulants. Collectively, these studies show a persistence of medication effects over time. Over 24 months of treatment, children with ADHD continued to respond well to MPH treatment, with no sign of a diminution of the drug’s efficacy. Domains of greatest improvement differ, with one study (Gillberg et al., 1997) showing greater effects at home and another (Schachar et al., 1997) showing bigger improvements at school. The mean total MPH daily doses reported during these 3 large-scale, randomized trials ranged from 30 to 37.5 mg/day. Dropping out was associated with lack of efficacy in the placebo condition or to the persistence of side effects.
Narcolepsy
MPH, DEX, and PEM have all been shown to significantly reduce daytime sleepiness in patients with narcolepsy (Mitler and Hajdukovic, 1990). Total daily doses in these studies were 60 mg for MPH and DEX and 112.5 mg for PEM. No studies of stimulant effectiveness have been done in children with narcolepsy, probably because the disorder is rarely diagnosed in the pediatric population.
Stimulants in the medically ill
Stimulants have been used to treat apathy and depression in medically ill patients, but dosages should be approximately one half the starting dose for ADHD and should be titrated slowly, with careful monitoring for side effects. Stimulants may be used in patients with apathy and depression secondary to medical illness (Frierson et al., 1991; Rosenberg et al., 1991). Yee and Berde (1994) used MPH (mean dose 14.6 mg/day) in 11 adolescent patients with cancer who were receiving large doses of opioid analgesics for pain. Although one patient developed hallucinations, 5 other patients showed increased attention and improved social interactions.
Stimulants also help reduce apathy or depression in seriously ill adult patients. A double-blind crossover trial of MPH showed it to be superior to placebo in reducing Hamilton Depression Scale scores in 16 depressed, medically ill patients (mean age 72.3 years) (Wallace et al., 1995). MPH was also superior to placebo in reducing depression and enhancing independent functioning in 21 poststroke patients (Grade et al., 1998). Although no controlled data exist, stimulants have been recommended for treatment of the disinhibited behavior that often occurs after head injury or in dementing illnesses (Gualtieri, 1991).
Treatment of adults with adhd
A majority of children diagnosed with ADHD may go on to meet DSM-IV criteria for ADHD in adult life (NIH Consensus Statement, 1998; Spencer et al., 1995b, 1996b). Prospective follow-up studies have shown that ADHD signs and symptoms continue into adult life (American Psychiatric Association, 1994). Adults with concentration problems, impulsivity, poor anger control, job instability, and marital difficulties seek help for problems they believe to be the manifestation of ADHD in adult life. Parents of children with ADHD may decide that they themselves have the disorder during an evaluation of their children (Ratey et al., 1992).
Determining whether an adult has ADHD and whether he/she is likely to benefit from stimulants requires a complete psychiatric evaluation, with particular focus on core ADHD symptoms starting in childhood. Because of the high rate of comorbid substance abuse, a detailed history of drug and alcohol use must be undertaken. One may consider obtaining a urine drug screen (Wilens et al., 1994a). In addition, information should be obtained from a spouse (or significant other), parent, or friend. Adults with ADHD often have notoriously poor insight and underestimate the severity of their ADHD symptoms and resulting impairments. A medical history, physical examination, and screening laboratory tests are useful in ruling out medical conditions that might masquerade as ADHD. Other conditions in the differential are bipolar disorder, depression, Axis II personality disorders, learning disabilities, narcolepsy, and undiagnosed borderline intellectual functioning. Structured rating scales have been found to be useful. These include the Wender Parent’s Rating Scale and the Wender Utah Rating Scales (Wender et al., 1981), the Brown Attention-Deficit Disorder Scale for Adults (Brown, 1996), and the Conners Adult ADHD Rating Scale.
Indications
The following conditions are indications for treatment with stimulant medication:
| • | ADHD without comorbid conditions. This includes all three subtypes of ADHD and ADHD, not otherwise specified (NOS). | ||||
| • | ADHD with specific comorbidities (oppositional defiant disorder, conduct disorder, anxiety disorder, and learning disorders). ADHD with certain Axis I anxiety disorders (separation anxiety disorder, generalized anxiety disorder, and social phobia) may be treated with stimulants. | ||||
| • | Narcolepsy. Patients with this disorder exhibit irresistible attacks of sleep that occur daily over at least a 3-month period. | ||||
| • | Apathy due to a General Medical Condition. Evidence for this is less, but it is used clinically. Individuals who have suffered brain injury due to trauma or a degenerative neurological illness often exhibit symptoms of inattention and impulsivity quite similar to ADHD. There is less evidence for this application of stimulant treatment, but stimulants and direct dopamine agonists have been used by clinicians. If the illness or trauma occurred after age 7 years, patients would not meet criteria for ADHD. Some patients with Alzheimer’s disorder or other dementing illnesses also exhibit impulsivity and inattentive behavior. Although no controlled trials exist documenting the effectiveness of stimulants in these conditions, clinical experience suggests that stimulants are helpful in reducing impulsive behaviors in some of these patients. Doses of the psychostimulants for these conditions are typically lower than those used in the treatment of ADHD. | ||||
| • | Adjuvant Medical Uses of Stimulants to treat severe psychomotor retardation. Evidence from controlled trials for this use is minimal, even though clinicians use stimulants to treat some severely medical ill patients who develop severe psychomotor retardation. This may be secondary to the illness itself, to the sedative effects of pain medication, or to toxic effects of the agents used to treat the primary illness (i.e., chemotherapy for cancer). Case reports suggest that low doses of stimulants may enable these patients to be more alert, eat better, and have a higher energy level. | ||||
Contraindications
The package insert for each stimulant medication is reproduced in full in the PDR (2000). Included are contraindications, warnings, and precautions. Some contraindications are stronger than others. For the psychostimulants, most of their listed contraindications have been found to present only minimal problems. Conversely, the package inserts fail to mention psychosis, which is probably a true contraindication. As a result, the FDA-approved package inserts do not serve as accurate guidelines for practitioners who choose to use stimulant medication. Contraindications relevant to clinical practice include:
| • | Concomitant use of MAO inhibitors. MAO inhibitors must not be used with stimulants. Severe hypertension will result, and there is a risk of a cerebrovascular accident. | ||||
| • | Psychosis. Stimulants are a known psychotomimetic for individuals with schizophrenia, so stimulants should not be used in patients with an Axis I diagnosis of schizophrenia, psychosis NOS, or manic episodes with psychosis. | ||||
| • | Glaucoma. There are suggestions that any sympathomimetic, including stimulants, may increase intraocular pressure. | ||||
| • | Existing liver disorder or abnormal liver function test results. PEM should not be used in patients with preexisting liver disease or abnormal liver function tests. The hematoxicity of PEM has been well documented. | ||||
| • | Drug dependence. A “black box” warning in the MPH, DEX, and AMP package inserts warns against using the medication in patients with a history of recent stimulant drug abuse or dependence. Patients who have histories of using or abusing other substances, such as cigarettes, alcohol, opiates, benzodiazepines, or sedatives, however, may have stimulants given to treat their ADHD. Even a history of abuse of stimulants may not represent an absolute contraindication. Of course, such patients must be monitored even more carefully than would otherwise be the case. | ||||
Other contraindications in the package insert have not been supported by data from recent randomized controlled trials. These include:
| • | Motor tics. Controlled studies have not found that MPH worsens motor tics in Tourette’s syndrome (Castellanos et al., 1997; Gadow et al., 1995), nor does it increase motor tics in children with ADHD without Tourette’s (Law and Schachar, 1999). It is possible to miss drug-related tics in group average data because of the noise associated with tic variability, but identifying a increasing dose-increasing tic-frequency relationship can be confirmative. Data from one study suggested that tic severity was worse with AMP than with MPH (Castellanos et al., 1997). | ||||
| • | Depression. Stimulants can produce dysphoria in vulnerable patients. For example, children treated with stimulants have been reported to become tearful and show tantrums when the effects of the medication wear off. The physician should be cautious in prescribing stimulants to a patient with an unstable mood disorder. Some ADHD patients with depressive signs resolve their secondary depression when their academic, behavioral, and social problems abate with stimulant treatment. | ||||
| • | Anxiety disorder. Children with comorbid anxiety disorder improve on MPH (Diamond et al., 1999; MTA Cooperative Group, 1999b). | ||||
| • | Seizure disorder. MPH in high doses may cause seizures in adults (Weiner, 1991). Children and adolescents with preexisting seizure disorders should be stabilized on anticonvulsants before treatment with stimulants. Once the seizures have been stabilized, treatment with stimulants can begin. | ||||
| • | Fatigue states. No evidence exists that stimulants worsen fatigue states. In fact, fatigue is a common nonmedical target for stimulants in the military. | ||||
| • | Children under age 6 (package insert only). As noted elsewhere in these parameters, there have been 7 double-blind studies involving 241 preschoolers with ADHD showing that MPH has good efficacy, with somewhat higher rates of adverse effects than reported in older children. Many of these studies were published after this warning first appeared in the package insert for MPH. Even so, more safety and efficacy information is needed before the treatment of preschoolers with MPH acquires the status of an evidence-based treatment. Paradoxically, AMP and DEX have been approved by the FDA for use in children as young as 3 years old, even though there are no published controlled data showing safety and efficacy. | ||||
Use of stimulants
Once the clinician and family have agreed to stimulant treatment, several steps must be planned. The parent first should be educated about the natural course of the disorder and the benefit-to-risk ratio of the medication treatment. Then comes the choice of medication. The literature does not help the clinician choose the best stimulant drug for an individual patient. Group studies of psychostimulants—MPH, DEX, and AMP—generally fail to show significant differences between DEX or AMP and MPH (Arnold, 2000). Conversely, there are large individual differences in response to different drugs and doses (Arnold et al., 1978; Elia et al., 1991). Therefore, the best order of their presentation for a particular patient is unknown. MPH, DEX, or AMP may be used first, on the basis of the inclinations of the physician and the parent.
The physician then needs to decide on a starting dose and a titration regimen. Published drug studies may not help the clinician select a dose for a particular patient, because studies do not report individual dose-response curves. In fact, most published studies treat all patients with the same stimulant medication dose, adjusted for the patient’s weight. Single-subject designs with rapid alternating drug conditions and multiple repeats yield the most reliable information on drug response but are often most difficult to implement (Kutcher, 1986; Pelham and Milich, 1991).
The research literature suggests two divergent methods for picking a starting dose of MPH for a particular child with ADHD: the weight-adjusted method and the fixed dose method. The weight-adjusted method allows the standardization of drug administration for different-size children. This method became popular after the publication of a seminal article on stimulant dosing (Sprague and Sleator, 1977) that reported dissociation between the cognitive and behavioral effects of MPH. The best cognitive test performance occurred at a lower weight-adjusted dose (0.3 mg/kg), whereas the best behavioral response was found at a higher dose (1.0 mg/kg). Unfortunately, few studies have been able to replicate Sprague’s work. One attempt found little correlation between weight-adjusted MPH doses and the reduction of ADHD symptoms (Rapport et al., 1989). Weight-adjusted dose ranges (0.3 or 0.8 mg/kg per dose) may restrict a titration trial for some small children, who require higher MPH doses to treat their ADHD symptoms. Current research does not uniformly support titrating with weight-adjusted doses.
Furthermore, the weight-adjusted titration method is problematic in office practice. Standard MPH tablets are unscored, so the fractional doses (e.g., 0.3 mg/kg) demanded by this method require the pills to be cut, resulting in pill fragments of unknown strength.
The alternative method uses fixed doses or whole or half MPH pills during titration. Total daily doses are increased through the 10 to 60 mg range until the child shows improvement or side effects. This escalating-dose, stepwise-titration method using whole pills reflects typical practice in the United States. The fixed-dose titration method, however, may expose small children to high doses of MPH, possibly resulting in untoward side effects.
Predicting drug response in an individual child is difficult. Pretreatment patient characteristics (young age, low rates of anxiety, low severity of disorder, and high IQ) may predict a good response to MPH for some children (Buitelaar et al., 1995). Yet most research shows that no neurological, physiological, or psychological measures are reliable predictors of response to psychostimulants (Pelham and Milich, 1991; Zametkin and Rapoport, 1987). Once a child responds, there is no universally agreed-upon criterion for how much the symptoms must change before the clinician stops increasing the dose.
Furthermore, there is no “gold standard” for the best outcome measure to use for guiding treatment. Some have advocated the use of “objective” tools, such as a CPT. Yet the CPT has a 20% false-positive rate and false-negative rate for identifying ADHD, and the utility of CPTs for adjusting dosages of medication has never been validated against classroom behavioral scores or academic performance. Clinicians should base decisions to change doses on scores on one of the many standardized, validated rating scales for assessing ADHD behavior.
Total daily MPH doses are increased through the 10 to 60 mg range until the child shows improvement or troublesome side effects. This escalating-dose, stepwise-titration method reflects typical practice in the United States, as described in clinical guides (Barkley et al., 1999; Dulcan, 1990; Greenhill, 1998a). One must take care to titrate slowly with small children and to stop or to reverse the dose increase when side effects occur. Experiencing unnecessary side effects may decrease the willingness of children or parents to use stimulants.
The clinician also must select the best time of day for drug administration and the dose given each time. Multiple doses of the immediate-release formulations must be given during the day, to cover school and the afternoon periods when homework is done. Even though the standard administration regimen is three times daily, one may have to adjust the exact timing of stimulant drug administration (Swanson et al., 1978), including the end-of-day dose’s timing and strength to minimize side effects (e.g., reduced appetite at dinner and delayed sleep onset). The clinician also must decide whether to pick a short- or long-acting stimulant formulation for maintenance treatment.
PhaseI: starting a stimulant medication
Treatment should be started with low doses of either MPH, DEX, or AMP (NIH Consensus Statement, 1998). Table 1 shows the titration schedules of the three stimulants. Patients are started on 5 mg of MPH or 2.5 mg of AMP/DEX; ideally, MPH is given after breakfast and lunch, with a third dose after school to help with homework and social activities. AMP/DEX may be started once daily in the early morning, with a noon dose added if it does not last through the school day. Evidence exists that increasing the morning dose of AMP may extend its duration of action (Pliszka et al., 2000a,b; Swanson et al., 1998a). If there is no improvement in symptoms, the dose may be increased in the following week. For children with ADHD, rating scales should be obtained from teachers and parents. For adults with ADHD, symptom and side-effect ratings can be collected from the patient and significant other before each dose increase. These ratings can be obtained through phone contact. Clinicians may stop titration upward when, in their clinical judgment, symptoms have resolved and impairment has been diminished. Different target symptoms may require different doses, so the clinician and family should prioritize which symptoms are to be chosen as a basis for titration.
Alternatively, a clinician may give a patient a “forced titration” trial—that is, the patient takes all four dosages of stimulant (5, 10, 15, 20 of MPH or 2.5, 5, 7.5, 10 of DEX/AMP), with each dose condition lasting 1 week. The total daily MPH dose range during titration for children weighing less than 25 kilograms in the MTA study reached up to 35 mg, although this could be exceeded if the child showed more room for improvement at that dose (Greenhill et al., 1996). Similar caution should be exercised for DEX/AMP for these small children, but no recommended maximum doses have yet been suggested. At the follow-up visit, rating scales from all 4 weeks are examined, along with reports of side effects, and the clinician selects the dose that produced the most benefit with the fewest side effects.
Adults or older adolescents may be started on doses of 5 mg of MPH, DEX, or AMP, with titration upward in 5- to 10-mg intervals each week until symptoms are controlled. Maximum daily doses for older adolescents and adults generally are similar to those for school-age children, with some adult patients treated with total daily doses of up to 1.0 mg/kg of MPH or 0.9 mg/kg of DEX/AMP, or 65 mg for MPH and 40 mg for DEX/AMP (Spencer et al., 1994). These higher total daily doses may be reached because adults need more dosings to cover a longer day. If the patient is taking larger doses, the clinician should clearly document that such symptoms could not be controlled at lower doses and the higher doses are not producing side effects (weight loss, blood pressure increase, or agitation). The patient should be monitored for signs of tolerance.
Phase2: alternative stimulant
It has been shown that approximately 70% of children with ADHD respond to either DEX or MPH alone. Nearly 90% will respond if both stimulants are tried (although some may have unacceptable side effects) (Elia et al., 1991). If a child fails to respond to the first stimulant tried or has moderate to prohibitive side effects (Table 2), the child can be switched to an alternative stimulant.
Sustained-release stimulants
For many years, the only long-acting preparations of DEX and MPH have been the DEX Spansule and MPH-SR. Patients have typically been started on the immediate-release preparation, with a later option of converting to the long-acting form. The morning and noon doses are added together; this gives the required dose of MPH-SR20. For example, if a patient were on MPH 10 mg in the morning and at noon, he/she would take 20 mg of MPH-SR20 in the morning. For the DEX Spansules, the morning and noon doses of DEX are added to yield the dose of the spansule.
It has become common practice to combine short-acting MPH with MPH-SR20 to increase efficacy and duration of effect and allow more flexible dosing. For instance, a child on 15 mg of MPH in the morning and at noon might be switched to 20 mg of SR in the morning and 5 mg of MPH-IR in the morning and at noon. If the dose of immediate release is taken before school, the medication starts working before the first class, whereas the SR-20 taken at the same time first begins to work during the middle of the morning. This strategy has been used to smooth out the day-long response by eliminating breakthrough ADHD symptoms. If an additional short-acting MPH tablet is then added midday, however, this defeats one of the purposes of the MPH-SR20 (to avoid in-school dosing).
Use of pemoline
Postmarketing surveillance revealed altered liver function tests in 44 children treated with PEM either short- or long-term (Berkovitch et al., 1995). More important, since the drug was introduced, 13 children experienced total liver failure, 11 resulting in death or transplantation within 4 weeks of failure. This rate is 4 to 17 times that expected in the normal population. As a result, PEM has now been listed as an alternative treatment one would select only after three or more stimulants have failed to be tolerated. The manufacturer further suggests that the drug be discontinued if no symptomatic improvement occurs within 3 weeks after the medication has been titrated to a clinically relevant dose (e.g., total daily dose of 56 mg/day). Parents must sign a written informed consent that clearly states that the child is at risk of liver failure and death and must comply with biweekly blood tests.
PEM treatment must be preceded by baseline liver function tests, specifically serum alanine aminotransferase (ALT). If ALT rises to twice normal values, PEM must be stopped. This test is followed biweekly, subjecting children to the pain and inconvenience of venipuncture. Patients may remain on PEM if they have failed all other agents and have maintained good symptom control on the drug. Recent psychopharmacology algorithms advise clinicians to try all three stimulants (MPH, DEX, and AMP) before turning to the antidepressants (bupropion or tricyclics) but do not include PEM (Fig. 1).
Although well-controlled trials of PEM documenting its efficacy outnumber the controlled trials of nonstimulant medications, PEM should be considered only as a last resort for those children who fail two stimulants (MPH and DEX or AMP), also fail the use of an antidepressant medication, and whose parents fill out the consent form now listed in the package insert. Physicians must inform families of the risk of severe hepatic failure. If liver function tests (aspartate aminotransferase, ALT, alkaline phosphatase) are within the normal range, a physician may consider standard PEM dosing. PEM is given as a single oral morning dose; if ADHD symptoms return in the afternoon, a second daily dose may be given. Children unresponsive to 56 mg can be titrated in 18.75- or 37.5-mg increments every 3 days to a maximum dose of 112.5 mg/day. Some children prefer the chewable, strawberry-flavored, 37.5-mg tablet.
Drug–drug interactions
Patients on MAO inhibitors are likely to develop hypertensive crises if given a stimulant. Drug-drug interactions do not occur, however, between stimulants and other antidepressants. Warning statements included in stimulant package inserts have been based on in vitro studies and anecdotal reports of increased tricyclic antidepressant (TCA) serum levels during combined treatment. More recent work includes a naturalistic study that showed no change in desipramine pharmacokinetics when stimulants were added (Cohen et al., 1999). Another prospective controlled study demonstrated that the isoenzyme CYPD2D6 does not play a prominent role in the metabolism of MPH in vivo, suggesting the lack of a clinical significant interaction with TCAs (DeVane et al., 2000). Even so, clinicians should be cautious in combining the drugs. A recently published single case report of a 10-year-old boy with ADHD who died of cardiac arrhythmia while being treated with 10 mg of DEX and 6.9 mg/kg per day of imipramine should be reviewed with the parents before this combination is started (Varley, 2000).
A selective serotonin reuptake inhibitor (SSRI) may be added to MPH for treating a child with ADHD and comorbid depression. This has been recommended by those constructing algorithms for the selection of drug treatments for ADHD children comorbid for depression (Pliszka et al., 2000a,b), although there are no controlled trials to support this. SSRIs are metabolized in the liver, whereas 80% of the metabolism of MPH is extrahepatic. This explains why no interactions have been reported for the combination.
Other drugs interact weakly with MPH. MPH inhibits the metabolism of certain anticonvulsants, so children on both may develop more side effects. MPH also interacts with guanethidine to produce paradoxical hypotension.
Rating forms
There are many rating scales for assessing the symptoms of ADHD. The clinician should select one of these scales—preferably one with age- and gender-specific norms—and use it to gather information on the patient before initiating stimulant treatment and after each major dose adjustment. Lack of teacher or parent cooperation may make the use of these scales difficult, but the clinician should make the effort and document the reason the scale could not be obtained. Table 3 shows the common scales in clinical use.
Age-specific use of stimulants
Preschool children
Eight published randomized controlled trials in preschool-age children attest to the robust efficacy of MPH. No controlled information is available, however, on dosing, long-term effects on development, or the patient characteristics associated with response.
Validity of the ADHD Diagnosis in the Preschool Age Range.
Disruptive behaviors suggestive of ADHD have been identified in children ages 3 to 5 years (the preschool period) (Campbell and Ewing, 1990). The lack of controlled prospective follow-up data on these symptoms and the paucity of developmentally appropriate examples in the DSM-IV criteria, however, make the diagnosis of ADHD in this age group more difficult. The differential diagnosis of ADHD in a preschooler includes adjustment disorders, other Axis I disorders that may show overactivity, impulsivity and poor attention (e.g., pervasive developmental disorder), and the normal high energy and exuberance of a 3-year-old. Oppositional behaviors are normally more common in 3-year-olds than 6-year-olds. ADHD is best identified when the child is asked to do sedentary tasks requiring sustained attention in a structured classroom setting, a situation not often experienced by preschoolers. Despite their young age, these children were identified by parents, teachers, and clinicians as seriously impaired.
Medication Treatment Studies of Preschool Children With ADHD.
MPH prescriptions for children in the preschool age range have increased 3-fold between 1991 and 1995, with 1.2% of the preschool population now estimated to be on MPH (Zito et al., 2000). There is a relative absence of information for preschool-age children compared with school-age children regarding MPH pharmacokinetics, pharmacodynamics, peak and duration of behavioral effects, interaction between drug and the developing brain, guidelines for dose response, and side effects related to short- and long-term exposure to stimulants.
Since 1975, there have been eight double-blind placebo-controlled trials of MPH in preschoolers with ADHD, involving 241 subjects (Barkley, 1988; Barkley et al., 1984; Cohen et al., 1981; Conners, 1975; Firestone et al., 1998; Handen et al., 1999; Mayes et al., 1994; Montieiro-Musten et al., 1997; Schliefer et al., 1975). All but two of these studies showed MPH to be superior to placebo, although some studies showed higher rates of side effects than are seen in studies with school-age children. One placebo-controlled study observed the following rate (compared with placebo) of side effects: irritability (26%), decreased appetite (20%), lethargy (19%), abdominal symptoms (12%), stereotypies (6%), and headaches (4%) (Mayes et al., 1994). Most published studies of MPH treatment of preschoolers use crossover designs to treat referred samples of children with ADHD but do not report the carryover effect or test for period by treatment interactions. The doses used were no greater than 0.5 mg/kg per dose, a narrower dosing range than the 0.3- to 0.8-mg/kg per dose range used in older children (Greenhill, 1998b). Doses were given once daily or twice daily, not the 3 times daily now used for school-age children. Trials were short in duration, with 5 of the 7 studies lasting 3 weeks or less. Methods varied for diagnosis, baseline or placebo conditions, and raters. Most had no input from teachers.
Generally, practitioners have to face two issues in prescribing for preschool children. The first is the starting dose. There are no published guidelines for starting doses for the age group. A 6-site NIMH trial of MPH in preschoolers is using starting doses of 1.25 mg tid.
Another treatment challenge is teaching preschoolers to swallow pills by use of behavioral training (Arnold et al., 1997). Alternatively, parents crush the pill in applesauce or find a pharmacy that will prepare a liquid suspension of the stimulant. Such preparations may not have the same absorption characteristics as the standard tablet.
Adolescents
Adolescents present other challenges for the prescriber. Adolescents are able to report the onset of stimulant action and may be able to detect the benefits of lengthened attention span. Although some may find the stimulant treatment of their ADHD supportive, others may rebel against frequent administration, trips to the school nurse, and feelings of dysphoria. Compliance with pill taking is no longer the total responsibility of the parent and teacher. It is important to work on medication management directly with the adolescent as well as with the parent. Longer-acting preparations may be more indicated for this population so as to maintain privacy in school. Concerta, a long-duration MPH preparation that is resistant to diversion (cannot be ground up or snorted), is well suited for treatment of adolescents. This long-acting preparation can be given once in the morning at home and an immediate-release stimulant given once in the afternoon before homework.
Treatment of adults with ADHD
Controlled studies of stimulant treatment have been conducted with more than 200 adult subjects with ADHD (Gualtieri et al., 1981; Mattes et al., 1984; Spencer et al., 1995a; Wender et al., 1981). Different pharmacological treatment strategies have been applied to ADHD in adults, with varying success. Studies of stimulant-treated adults have produced divergent rates of drug efficacy (23% to 75%) (Wilens and Biederman, 1992). This variability may be the result of low stimulant dosages, the high rate of comorbid disorders, and/or different diagnostic criteria. Some studies have used self-reported outcome measures, even though adult ADHD patients are unreliable reporters of their own behaviors. Outcomes range from minimal benefit from MPH (Mattes et al., 1984) to robust effects (Wender et al., 1985). Using higher doses, Spencer and his colleagues reported that the response to a total daily dose of 1 mg/kg of MPH in 23 adults with ADHD was independent of gender, comorbidity, or family history of psychiatric disorders (Spencer et al., 1995b). Seventy-eight percent showed improvement on MPH versus 4% who responded to placebo. Treatment was generally well tolerated; side effects included loss of appetite, insomnia, and anxiety. Other drugs that have been reported anecdotally to be beneficial include fluoxetine (Sabelsky, 1990), pargyline (Wender et al., 1994), bupropion (Wender and Reimherr, 1990), and the MAO inhibitor selegiline (Ernst et al., 1995).
Stimulant medications may be used to treat carefully evaluated adults with ADHD (DuPaul and Barkley, 1990; Wender et al., 1994). These include MPH, 5 to 20 mg tid, and DEX, 5 mg tid to 20 mg bid. Of particular concern is the danger of prescribing psychostimulants for adults with comorbid substance abuse disorder.
Treatment of ADHD with comorbid disorders
ADHD may be comorbid with a variety of psychiatric disorders (Biederman et al., 1991; Pliszka, 1992), including but not limited to depression or anxiety, tic disorders, oppositional defiant disorder (ODD), conduct disorder, and/or severe aggressive outbursts.
Depression/anxiety disorders
If major depressive disorder (MDD) is the primary disorder or if MDD is accompanied by very severe symptoms (psychosis, suicidality, or severe neurovegetative signs), the MDD should be the focus of treatment. If the MDD is less severe or is not primary, however, then there is an advantage to performing a stimulant trial first. Onset of stimulant treatment is rapid, so that the physician can quickly assess whether the ADHD symptoms have remitted. The reduction in morbidity caused by the ADHD symptoms can have a substantial impact on the depressive symptoms. After the stimulant trial, the physician can evaluate the depressive symptoms. If the depressive and ADHD symptoms both have remitted, no other changes may be necessary in the treatment plan. Conversely, if the ADHD symptoms have responded but the depressive symptoms remain severe, psychotherapeutic treatment, such as cognitive behavioral therapy or interpersonal therapy, or an antidepressant should be considered.
No data support a single antidepressant to treat both ADHD and MDD. Although bupropion and tricyclics have proven antidepressant activity in adults, their utility in treating pediatric depression has not been established. They are second-line agents, at best, for treating ADHD.
Data on the treatment of comorbid anxiety disorder in ADHD children are more easily available. Early work showing that ADHD children with comorbid anxiety had a less robust response to stimulants has not been replicated in later, more extensive clinical trials (Diamond et al., 1999; MTA Cooperative Group, 1999a). In fact, differences between treatment groups in the MTA study increased if the subjects with ADHD had comorbid anxiety disorder. Thus, the clinician should proceed with a stimulant trial. If the stimulant improves the ADHD symptoms but the anxiety symptoms remain problematic, the clinician may pursue a psychosocial intervention for the anxiety. If the anxiety does not respond to nonpharmacological treatment or is severe, the clinician may consider adding an SSRI to the stimulant. The clinician should consult the AACAP parameters on the treatment of anxiety disorders for further information on this topic (American Academy of Child and Adolescent Psychiatry, 1997b).
Tic disorders
Recently, a number of double-blind placebo-controlled studies have examined the effects of stimulant medication in children with comorbid ADHD and tic disorders (Castellanos et al., 1997; Gadow et al., 1995; Law and Schachar, 1999). These studies showed that the stimulants are highly effective in the treatment of ADHD in these patients and that in the majority of patients tics do not increase. With proper informed consent, a trial of a stimulant could be undertaken in children with comorbid ADHD and tic disorder. If tics worsen markedly, the physician would move to an alternative stimulant. If tics do not increase and the ADHD symptoms respond, however, then the child may remain on the stimulant. The tics may remain problematic. If so, the physician may consider a number of agents to combine with the stimulant. Such α-agonists as clonidine or guanfacine may be tried first.
Conduct disorder and aggression
Severe aggressive outbursts are seen in some ADHD children, particularly those with comorbid conduct disorder. A number of studies have shown that antisocial behaviors in school-age children, such as stealing and fighting, can be reduced by stimulant treatment (Hinshaw et al., 1992; Klein et al., 1997; Murphy et al., 1986). The physician should assess the effectiveness of the stimulant in reducing antisocial behavior. If aggressive outbursts remain problematic despite the attenuation of the ADHD symptoms, then mood stabilizers (lithium or divalproex sodium) or an α2-agonist may be considered for addition to the stimulant medication (Campbell et al., 1984).
If aggression in children with ADHD is pervasive, severe, and persistent and is an acute danger to themselves and others, it may be justifiable to add an atypical neuroleptic, such as 0.5 mg qd risperidone, to the stimulant. Risperidone has been shown to decrease aggression in children and adolescents with pervasive developmental disorders and conduct disorder (Frazier et al., 1999; McDougle et al., 1997). A recent report (n=20) suggests that 6 weeks of divalproex treatment for adolescents (ages 10 to 18 years) with explosive temper and mood lability has been successful in showing a 70% reduction from baseline in scores on the Modified Overt Aggression Scale and the SCL-90 anger-hostility items (Donovan et al., 2000).
Can the response to stimulants be augmented by other psychotropics?
The above recommendations involve adding a second medication to treat symptoms that are comorbid with the ADHD symptoms. There has been speculation among experienced clinicians for many years that adding an antidepressant such as a tricyclic or bupropion can further enhance the effect of the stimulant on the ADHD symptoms themselves. One case report of leukopenia was reported in a child treated with a combination of imipramine and MPH for 4 months, but the doses were not specified (Burke et al., 1995). The child was also mentally retarded and highly aggressive.
Desipramine and MPH were used alone and in combination in two double-blind, placebo-controlled crossover studies (Pataki et al., 1993; Rapport et al., 1993). This was an inpatient study in which careful electrocardiographic (ECG) and TCA-blood-level monitoring were carried out. The mean daily dosage of desipramine was 4.04 mg/kg per day, with a range of 2.4 to 6.1 mg/kg per day. Desipramine plasma levels ranged from 121 to 291 ng/mL. The MPH dose range was 10 to 40 mg/day. The 16 subjects were 7 to 12 years old, most had comorbid mood disorders and ADHD, and all were severely ill inpatients. Unfortunately, no measures of clinical response were included, only computerized measures of attention and impulsivity. The combination was superior on some measures and inferior on others compared with either medication alone. Side effects such as nausea, dry mouth, and tremor were twice as common on combined desipramine and MPH relative to either drug alone, but they were mild. The authors concluded, “The main implication of the study is during the 3 to 4 month protocol, there was no clinical evidence of unique or serious side effects in combining desipramine and MPH beyond those attributable to desipramine alone” (Rapport et al., 1993). The use of TCAs in children with ADHD and in depression has fallen off because of the association with sudden death in five children, as reported a decade ago (Biederman, 1991; Popper and Zimnitzky, 1995; Varley and McClellan, 1997).
Clonidine, the α2-agonist, has been combined with MPH to reduce aggression, to provide better control of ADHD symptoms after the stimulant has worn off, and to counteract the insomnia that can occur with stimulants (Wilens et al., 1994b; Wilens and Spencer, 1999). A recent meta-analysis shows that clonidine alone may have some efficacy in the treatment of ADHD (Connor et al., 1999). Concern about this combination of stimulants and α2-agonists raised by the report of four deaths of patients reported on the FDA’s MEDWATCH surveillance network has worried clinicians and led to warnings about combining the two medications (Swanson et al., 1999b). There have been no further reports, however, and clinicians continue to use the combination. Because of the largely negative findings from routine ECG monitoring during treatment with this combination, ECGs at baselines are not advised. The rate of such side effects as bradycardia, hypotension, and hypertension appears to be in the rare to infrequent (less than 1/100) range.
If the combination is to be used, start with a half a clonidine tablet (0.05 mg) at bedtime and increase the dose slowly, never giving more than 0.3 mg/day. It needs to be given at bedtime for sleep or used 4 times daily if one wishes to control aggressive behavior. Before starting clonidine, one must take a full medical history of the patient and first-degree family members. A history of sudden death, repeated fainting, or arrhythmias in family members probably would rule out its use.
There are no studies of the combination of bupropion and stimulants in the treatment of ADHD or other psychiatric conditions. The PDR does not warn against interactions between bupropion and stimulants, and we could locate no case reports of side effects when these agents were combined. Nonetheless, clinicians should proceed with caution in combining these agents until further studies are available.
Monitoring treatment: drug discontinuation, frequency of visits
Once the child with ADHD is stabilized on stimulant medication, visits may be scheduled once a month. In the MTA study (MTA Cooperative Group, 1999a), once-monthly, 30-minute medication visits with the parent and child were found to result in significantly lower ratings of teacher and parent core ADHD symptoms compared with treatment as usual in the community. Compared with children assigned to the MTA’s community comparison group, children in the MTA’s medication management treatment arm were treated with doses 10 mg/day greater; had 3-times-daily dosing versus twice-daily dosing; started treatment with an intensive, 28-day, double-blind titration trial; received supportive counseling and reading materials; and had their dosage adjustments informed by monthly teacher consultation by the pharmacotherapist. Future prospective, controlled dismantling studies will be necessary to determine which of these elements contributed most to the success of the MTA medication management protocol.
For the practitioner, the monitoring plan should include regular visits. Although the stimulant’s schedule II status means no refills, practitioners can fill out prescriptions on a monthly basis, and sometimes once every 3 months, as in New York State. Children with stable responses to the stimulant and no comorbidities can be seen once every 3 or 4 months. Complex comorbidity or side effects may require monthly visits.
Should each stimulant-treated patient be discontinued from medication at least once a year? Parents and practitioners grapple with the advantages and disadvantages of drug discontinuation trials. The stimulants work only as long as they are given, so stopping the drug usually results in the rapid return of symptoms. Even so, many parents favor a period off drug, typically called a “drug holiday,” to deal with concerns about lack of weight gain and worries about possible long-term drug effects or to assess the continuing need for staying on medication. If this type of trial is to be run, it is best done at times other than when the child is scheduled for important school tests, is just starting the school year, or is involved in important social activities (e.g., summer camp).
Complications and side effects
Stimulant-related side effects reported for children with ADHD appear to be mild, short-lived, and responsive to dose or timing adjustments. Adverse drug reactions usually occur early in treatment and often decrease with dose adjustment. Double-blind, placebo-controlled studies report moderate side effects in 4% to 10% of children treated. Delay of sleep onset, reduced appetite, stomachache, headache, and jitteriness are the most frequently cited (Barkley et al., 1990). No additional delay in sleep onset was seen after a third, midafternoon dose of MPH was added to standard twice-daily dosing regimens (Kent et al., 1995). Some children experience motor tics while on stimulants, but the mechanism for this is unclear. Twenty-three controlled studies found no differences for these side effects among the stimulants, with only abdominal discomfort, sleep delay, and headache being reported more often for stimulant than for placebo in 2 or more of the 23 controlled trials (McMaster University Evidence-Based Practice Center, 1998).
Rarely, children have been reported to display cognitive impairment or perseverative behaviors, but this usually responds to a decrease in dose. Children also have shown mood disturbances or, very rarely, psychosis or hallucinosis.
Staring, daydreaming, irritability, anxiety, or nailbiting may decrease with increasing stimulant dose. No consistent reports of behavioral rebound, motor tics, compulsive picking of nose or skin, dose-related emotional or cognitive constriction, or dose-related growth delays have been found in controlled studies (Spencer et al., 1996a). This low rate of stimulant side effects, however, emerges from short-term trials (Mayes et al., 1994).
Concern has been raised about stimulant-related growth delays. Small weight decrements are reported during short-term trials (Gittelman-Klein et al., 1988), but prospective follow-up into adult life (Manuzza et al., 1991) has revealed no significant impairment of height attained. Furthermore, the growth rate delays attributed to medication may be a developmental artifact associated with the disorder. ADHD children in the MTA study treated with chronic stimulants, however, showed significant decrements in rates of weight acquisition compared with ADHD children randomized to a nonmedication treatment (Greenhill and MTA Cooperative Group, 1999). Changes in rates of height acquisition in the MTA study differed only minimally between the groups during the 14-month treatment period and did not reach clinical significance.
With an estimated 3 million children and adolescents in the United States taking stimulants daily, the occurrence of serious “side effects” in children and adolescents is in the range of very rare (less than 1/10,000). The small number of incidents makes it difficult to determine whether these very rare but serious side effects are related to the chronic use of stimulants in children. If there were such a relationship, many more occurrences would be expected, with a prescribing base rate in the millions. The uncertainty surrounding these tragic events makes it imperative that methodologies for carefully monitoring children on long-term treatment—a decade or more—to determine whether rare but serious side effects do occur that are related to the stimulant medication treatment.
Tactics for dealing with stimulant-associated side effects
Clinicians have used a variety of tactics to deal with side effects such as insomnia and appetite loss. These include:
| • | For appetite loss, the child can be given the stimulants with meals and a high-calorie drink or snack late in the evening, when the stimulant effects have worn off. | ||||
| • | For difficulty falling asleep, one must distinguish whether the delay in sleep onset is due to a side effect of the stimulant or from oppositionality related to the ADHD or to separation anxiety. First, lower the last stimulant dose of the day or move it to earlier in the day. To deal with the oppositional behavior, the clinician can help the parents implement a bedtime ritual (e.g., reading). | ||||
| • | For sadness, the clinician should reevaluate the diagnosis, reduce the dose, and change to sustained-release products (methylphenidate-SR, Concerta), because the peak of immediate-release stimulant may be causing more depressive effects. | ||||
| • | For behavioral rebound, one can overlap the stimulant dosing pattern, switch to longer-acting stimulants, combine immediate-release with sustained release, or add other medications (e.g., bupropion). | ||||
| • | For irritability, first evaluate when it occurs (if just after medication given, it may be a peak; if late in the afternoon, it may be rebound); reduce the dose. | ||||
Abuse potential of stimulant medications
Stimulant medications are classified as drugs of abuse by the Drug Enforcement Administration. In animal laboratory experiments, DEX, MPH, and AMP all show characteristics of abuse (e.g., self-administration, chosen in preference over food). Concerns are increasing about the abuse potential, because production and use of MPH increased 5-fold between 1986 and 1996. The increasing production and use have led to the following set of concerns (Goldman et al., 1998): ADHD children are at increased risk for drug use and abuse in adolescence; ADHD adolescents are being treated in increasing numbers with MPH; MPH may become increasingly abused or diverted or may serve as a gateway to other illicit drugs. Conversely, there have been two reports that ADHD adolescents treated with stimulants show lower rates of substance use disorder than ADHD adolescents not in treatment (Molina and Pelham, unpublished, 1999; Wilens et al., 1999). Parents and other family members may abuse the child’s stimulant medications, so it is important to ask whether anyone in the house has a problem with substance abuse.
Two considerations temper these concerns. One is the limited ability of the prescribed stimulants to induce euphoria by the oral route. PET scan studies have shown that oral MPH demonstrates markedly slower absorption, occupancy of the dopamine transporter, and decay than does intravenous cocaine; similarly, oral MPH does not induce euphoria (Volkow et al., 1995). Similarly, although MPH does appear in emergency room mentions in the Drug Abuse Warning Network, its mention rate is only 1/40th of the mention rate of cocaine (Goldman et al., 1998). Second, an increased risk of drug abuse and cigarette smoking is associated with childhood ADHD. The media have reported that college students use stimulants to enhance sports, studying, and paper-writing, rather than for euphoria. An unknown number of high school and college students have been reported to sell their stimulant medication or use it for recreational purposes by crushing and snorting the tablets.
The Drug Enforcement Administration has been supportive of a public program of education about the serious potential of stimulant drugs to be diverted and sold as drugs of abuse. They have encouraged local schools to keep the drugs in locked cabinets, to keep careful dispensing records, and never to use one child’s medication to treat another child. In general, it is better if the practitioner can avoid sending stimulant medications to an adolescent’s school to reduce the opportunity of drug diversion and to avoid peer ridicule.
The newer stimulant preparations, such as Concerta, are less prone to abuse and diversion than the immediate-release MPH tablets and are more suitable for adolescents with ADHD who are at risk for abusing their stimulant medications. With this drug, the medication need be given only once daily by the parents and not taken to school, where it could be given away or sold. Furthermore, the MPH in this extended-release caplet is in the form of a paste, which cannot be ground up or snorted.
Conflict of interest
As a matter of policy, some of the authors of this practice parameter are in active clinical practice and may have received income related to treatments discussed in these parameters. Some authors may be involved primarily in research or other academic endeavors and also may have received income related to treatments discussed in this parameter. To minimize the potential for this parameter to contain biased recommendations because of conflict of interest, the parameter was reviewed extensively by Work Group members, consultants, and AACAP members; authors and reviewers were asked to base their recommendations on an objective evaluation of the available evidence; and authors and reviewers who believed that they might have a conflict of interest that would bias or appear to bias their work on this parameter were asked to notify the AACAP.
Scientific data and clinical consensus
Practice parameters are strategies for patient management, developed to assist clinicians in psychiatric decision-making. This parameter, based on evaluation of the scientific literature and relevant clinical consensus, describes generally accepted approaches to assess and treat specific disorders or to perform specific medical procedures. The validity of scientific findings was judged by design, sample selection and size, inclusion of comparison groups, generalizability, and agreement with other studies. Clinical consensus was determined through extensive review by the members of the Work Group on Quality Issues, child and adolescent psychiatry consultants with expertise in the content area, the entire AACAP membership, and the AACAP Assembly and Council.
This parameter is not intended to define the standard of care, nor should it be deemed inclusive of all proper methods of care or exclusive of other methods of care directed at obtaining the desired results. The ultimate judgment regarding the care of a particular patient must be made by the clinician in light of all the circumstances presented by the patient and his or her family, the diagnostic and treatment options available, and available resources. Given inevitable changes in scientific information and technology, these parameters will be reviewed periodically and updated when appropriate.
| Measures Taken | ||||||
|---|---|---|---|---|---|---|
| Week | Dosage of Stimulant (mg) | Mode of Contact | ADHD Rating Scale | Side Effects | BP, Pulse, Ht, Wt | |
| Baseline | MPHa | DEXa/AMPb | Office visit | X | X | X |
| 1 | 5 | 2.5 | Visit | X | X | X |
| 2 | 10 | 5 | Visit or phone | X | X | |
| 3 | 15 | 7.5 | Visit or phone | X | X | |
| 4c | 20 | 10 | Visit | X | X | X |
| 5 | Further titration | Physician office visit | Review scales | X | ||
| Prohibitive (requires dose reduction or discontinuation) | Major (may require dose reduction, prohibits higher dose) | Minor (expected, tolerable) |
|---|---|---|
| Severe anorexia | Moderate anorexia | Mild anorexia |
| Severe insomnia (>1.5 hr) | Moderate insomnia (>1–1.5 hr) | Mild insomnia (>1 hr) |
| New, marked, severe tics | Fleeting new tics | Fleeting, negligible tics, causing no impairment |
| Severe, unrelenting headaches | Moderate headaches | Mild headaches |
| Intolerable GI cramps | Moderate GI cramps | Mild GI cramps |
| Severe picking at skin, nail-biting | Moderate picking at skin, nail-biting | Mild picking at skin, nail-biting |
| Severe anxiety | Moderate anxiety | Mild anxiety |
| Severe irritability, leading to aggression | Moderate irritability | Mild irritability |
| Severe depression not, preexisting | Moderate depression, not preexisting | Mild depression |
| Hallucinations | Questionable hallucinations | |
| “Zombie” all day | “Zombie” part of the day | Dull, tired listless |
| Pyschosis |
| Name of Scale | Reference |
|---|---|
| Conners Parent Rating Scale–Revised (CPRS-R) | Parent, adolescent self-report versions available (Conners, 1997) |
| Conners Teacher Rating Scale–Revised (CTRS-R) | Teacher (Conners, 1997) |
| Child Behavior Checklist (CBCL) | Parent-completed CBCL (Achenbach, 1991) and teacher-completed Teacher’s Report Form (TRF) |
| Home Situations Questionnaire–Revised (HSQ-R), School Situations Questionnaire–Revised (SSQ-R) | The HSQ-R is a 14-item scale designed to assess specific problems with attention and concentration across a variety of home and public situations. It uses a 0–9 scale and has test-retest, internal consistency, construct validity, discriminant validity, concurrent validity, and norms (n=581) available (Barkley, 1990b). |
| Conners Adult ADHD Rating Scales (CAARS) | Available in both long (66-item) and screening (18-item) versions (Conners, 1998) |
| Academic Performance Rating Scale (APRS) | The APRS is a 19-item scale for determining a child’s academic productivity and accuracy in grades 1–6 that has 6 scale points. Construct, concurrent, and discriminant validity data, as well as norms (n=247) available (Barkley, 1990a) |
| ADHD Rating Scale–IV (alternative to SNAP below) | The ADHD Rating Scale-IV is an 18-item scale using DSM-IV criteria (DuPaul, et al., 1998) |
| Children’s Attention Problems (CAP) | The CAP is a 2-factor (inattention and overactivity), 12-item teacher-rated scale for 6–16-year-olds developed by Edelbrock from the Teacher’s Report Form. It is convenient to use weekly to assess treatment outcome. It has internal consistency reliability information, construct validity, discriminant validity and concurrent validity, and norms (n=1,100) available (Barkley, 1990b) |
| IOWA Conners Teacher Rating Scale | The IOWA Conners is a 10-item scale developed to separate the inattention and overactivity ratings from oppositional defiance (Loney and Milich, 1982). It is useful in following treatment progress in children with both ADHD and oppositional defiant disorder. |
| Swanson, Nolan, and Pelham (SNAP-IV) and SKAMP Internet site ADHD.NET | The SNAP-IV (Swanson, 1992) is a 26-item scale that contains DSM-IV criteria for ADHD and screens for other DSM diagnoses. The SKAMP (Wigal et al., 1998) is a 10-item scale that measures impairment of functioning at home and at school. |
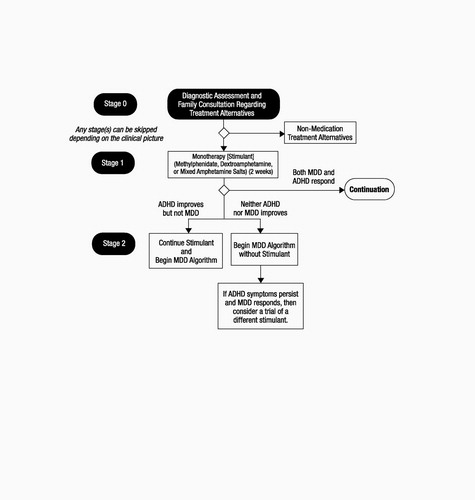
Figure 1. Medication Algorithm for Stimulants (Pliszka et al., 2000a). MDD=Major Depressive Disorder; ADHD=Attention-Deficit/Hyperactivity Disorder
Abikoff H, Gittelman R (1985), Hyperactive children treated with stimulants: is cognitive training a useful adjunct? Arch Gen Psychiatry 42:953–961Crossref, Google Scholar
Achenbach T (1991), Manual for the Child Behavior Checklist/4–18 and 1991 Profile. Burlington: University of Vermont Department of Psychiatry American Academy of Child and Adolescent Psychiatry (1997a), Practice parameters for the assessment and treatment of attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry 36:85S–121SGoogle Scholar
American Academy of Child and Adolescent Psychiatry (1997b), Practice parameters for the assessment and treatment of children and adolescents with anxiety disorders. J Am Acad Child Adolesc Psychiatry 36:69S–84SCrossref, Google Scholar
American Psychiatric Association (1994), Diagnostic and Statistical Manual of Mental Disorders, 4th edition (DSM-IV). Washington, DC: American Psychiatric AssociationGoogle Scholar
Angold A, Erkanli A, Egger H, Costello J (2000), Stimulant treatment for children: a community perspective. J Am Acad Child Adolesc Psychiatry 39: 975–983Crossref, Google Scholar
Angrist BM, Gershon S (1972), Psychiatric sequelae of amphetamine use. In: Psychiatric Complications of Medical Drugs, Shader RI, ed. New York: Raven, pp 175–199Google Scholar
Arnold L (2000), Methylphenidate vs amphetamine: comparative review. J Attention Disord 3: 200–211Crossref, Google Scholar
Arnold L, Abikoff H, Cantwell D et al. (1997), NIMH Collaborative Multimodal Treatment Study of Children With ADHD (MTA): design, methodology, and protocol evolution. J Attention Disord 2: 141–158Crossref, Google Scholar
Arnold LE, Christopher J, Huestis R, Smeltzer D (1978), Methylphenidate vs dextroamphetamine vs caffeine in minimal brain dysfunction. Arch Gen Psychiatry 35: 463–473Crossref, Google Scholar
Barkley R (1988), The effects of methylphenidate on the interactions of preschool ADHD children with their mothers. J Am Acad Child Adolesc Psychiatry 27: 336–334Crossref, Google Scholar
Barkley R (1990a), Child behavior rating scales and checklists. In: Assessment and Diagnosis in Child Psychopathology, Rutter M, Tuma AH, Lann IS, eds. New York: Guilford, pp 113–155Google Scholar
Barkley R, DuPaul G, Connor D (1999), Stimulants. In: Practitioner’s Guide to Psychoactive Drugs for Children and Adolescents, 2nd ed, Werry J, Aman M, eds. New York: Plenum, pp 213–241Google Scholar
Barkley R, McMurray M, Edelbroch C, Robbins K (1990), Side effects of MPH in children with attention deficit hyperactivity disorder: a systematic placebo-controlled evaluation. Pediatrics 86: 184–192Google Scholar
Barkley RA (1977), A review of stimulant drug research with hyperactive children. J Child Psychol Psychiatry 18: 137–165Crossref, Google Scholar
Barkley RA (1982), Hyperactive Children: A Handbook for Diagnosis and Treatment. New York: GuilfordGoogle Scholar
Barkley RA (1990b), Behavior rating scales. In: Attention-Deficit/Hyperactivity Disorder: A Handbook for Diagnosis and Treatment. New York: Guilford, pp 278–326Google Scholar
Barkley RA (1997), ADHD and the Nature of Self-Control. New York: GuilfordGoogle Scholar
Barkley RA, Karlsson J, Strzilecki E (1984), Effects of age and Ritalin dosage on mother–child interactions of hyperactive children. J Consult Clin Psychol 52: 750–758Crossref, Google Scholar
Battaglia G, Yeh S, O’Hearn E, Molliver M, Kuhar M, De Souza E (1987), 3,4-Methyoxydioxymethamphetamine and 3,4-methylenedioxyamphetamine destroy serotonin terminals in rat brain: quantification of neurodegeneration by measurement of [3H]paroxetine-labeled serotonin uptake sites. J Pharmacol Exp Ther 242: 911–916Google Scholar
Berkovitch M, Pope E, Phillips J, Koren G (1995), Pemoline-associated fulminant liver failure: testing the evidence for causation. Clin Pharmacol Ther 57: 696–698Crossref, Google Scholar
Biederman J (1991), Sudden death in children treated with a tricyclic antidepressant: a commentary. Biol Ther Psychiatry Newsl 14: 1–4Google Scholar
Biederman J, Newcorn J, Sprich S (1991), Comorbidity of attention deficit hyperactivity disorder with conduct, depressive, anxiety, and other disorders. Am Psychiatry 148: 564–577Crossref, Google Scholar
Birmaher BB, Greenhill L, Cooper T, Fried J, Maminski B (1989), Sustained release methylphenidate: pharmacokinetic studies in ADDH males. J Am Acad Child Adolesc Psychiatry 28: 768–772Crossref, Google Scholar
Bradley C (1937), The behavior of children receiving benzedrine. Am J Psychiatry 94: 577–585Crossref, Google Scholar
Bradley C, Bowen M (1941), Amphetamine (benzedrine) therapy of children’s behavior disorders. Am J Orthopsychiatry 11: 92–103Crossref, Google Scholar
Brown T (1996), Brown Attention Deficit Disorder Scales: Manual. San Antonio, TX: Psychological CorporationGoogle Scholar
Buitelaar J, Gary R, Swaab-Barneveld H, Kuiper M (1995), Prediction of clinical response to methylphenidate in children with attention deficit hyperactivity disorder. J Am Acad Child Adolesc Psychiatry 34: 1025–1032Crossref, Google Scholar
Burke MS, Josephson A, Lightsey A (1995), Combined methylphenidate and imipramine complication (letter). J Am Acad Child Adolesc Psychiatry 34: 403–404Crossref, Google Scholar
Campbell M, Small A, Green W (1984), Behavioral efficacy of haloperidol and lithium carbonate. Arch Gen Psychiatry 41: 650–656Crossref, Google Scholar
Campbell S, Ewing L (1990), Follow-up of hard-to-manage preschoolers: adjustment at age 9 and predictors of continuing symptoms. J Child Psychol Psychiatry 31: 871–879Crossref, Google Scholar
Carlson GA, Loney J, Salisbury H, Kramer JR, Arthur C (2000), Stimulant treatment in young boys with symptoms suggesting childhood mania: a report from a longitudinal study. J Child Adolesc Psychopharmacol 10: 175–184Crossref, Google Scholar
Castellanos X, Giedd J, Elia J et al. (1997), Controlled stimulant treatment of ADHD and comorbid Tourette’s syndrome: effects of stimulant and dose. J Am Acad Child Adolesc Psychiatry 36: 589–596Crossref, Google Scholar
Chan YP, Swanson JM, Soldin SS, Thiessen JJ, Macleod SM (1983), Methylphenidate hydrochloride given with or before breakfast, II: effects on plasma concentration of methylphenidate and ritalinic acid. Pediatrics 72: 56–59Google Scholar
Cohen LG, Prince J, Biederman J et al. (1999), Absence of effect of stimulants on the pharmacokinetics of desipramine in children. Pharmacotherapy 19: 746–752Crossref, Google Scholar
Cohen N, Sullivan J, Minde K, Novack C, Helwig C (1981), Evaluation of the relative effectiveness of methylphenidate and cognitive behavior modification in the treatment of kindergarten-aged hyperactive children. J Abnorm Child Psychol 9: 43–54Crossref, Google Scholar
Conners CK (1975), Controlled trial of methylphenidate in preschool children with minimal brain dysfunction. Int J Ment Health 4: 61–74Crossref, Google Scholar
Conners CK (1997), Conners’ Rating Scales-Revised. Toronto: Multi-Health SystemsGoogle Scholar
Conners CK (1998), Rating scales in attention deficit/hyperactivity disorder. J Clin Psychiatry 59(suppl 7):24–30Crossref, Google Scholar
Conners CK, Eisenberg L, Barcai A (1967), Effect of dextroamphetamine on children: studies on subjects with learning disabilities and school behavior problems. Arch Gen Psychiatry 17: 478–485Crossref, Google Scholar
Connor DF, Fletcher KE, Swanson J (1999), A meta-analysis of clonidine for symptoms of attention-deficit hyperactivity disorder. J Am Acad Child Adolesc Psychiatry 38: 1551–1559Crossref, Google Scholar
Cox BM (1990), Drug tolerance and physical dependence. In: Principles of Drug Action: The Basis of Pharmacology, Pratt WB, Taylor P, eds. New York: Churchill Livingstone, pp 639–690Google Scholar
DeVane C, Markowitz JS, Carson SW et al. (2000), Single-dose pharmacokinetics of methylphenidate in CYP2D6 extensive and poor metabolizers. J Clin Psychopharmacol 20: 347–349Crossref, Google Scholar
Diamond I, Tannock R, Schachar R (1999), Response to methylphenidate in children with ADHD and comorbid anxiety. J Am Acad Child Adolesc Psychiatry 38: 402–409Crossref, Google Scholar
Donovan SJ, Stewart JW, Nunes EV et al. (2000), Divalproex treatment for youth with explosive temper and mood lability: a double-blind, placebo-controlled crossover design. Am J Psychiatry 157: 818–820Crossref, Google Scholar
Dulcan M (1990), Using psychostimulants to treat behavior disorders of children and adolescents. J Child Adolesc Psychopharmacol 1: 7–20Crossref, Google Scholar
Dunnick J, Hailey J (1995), Experimental studies on the long-term effects of methylphenidate hydrochloride. Toxicology 103: 77–84Crossref, Google Scholar
DuPaul G, Barkley R, McMurray M (1994), Response of children with ADHD to methylphenidate: interaction with internalizing symptoms. J Am Acad Child Adolesc Psychiatry 33: 894–903Crossref, Google Scholar
DuPaul GJ, Barkley RA (1990), Medication therapy. In: Attention Deficit Hyperactivity Disorder: A Handbook for Diagnosis and Treatment, Barkley RA, ed. New York: Guilford, pp 573–612Google Scholar
DuPaul GJ, Power TJ, Anastopoulos AD, Reid R (1998), ADHD Rating Scales-IV: Checklists, Norms and Clinical Interpretation. New York: GuilfordGoogle Scholar
Eisenberg L, Lachman R, Molling P, Lockner A, Mizelle J, Conners C (1961), A psychopharmacologic experiment in a training school for delinquent boys: methods, problems and findings. Am J Orthopsychiatry 33: 431–447Crossref, Google Scholar
Elia J, Borcherding B, Rapoport J, Keysor C (1991), Methylphenidate and dextroamphetamine treatments of hyperactivity: are there true non-responders? Psychiatry Res 36:141–155Crossref, Google Scholar
Ernst M, Liebenauer L, Jons P, Murphy D, Zametkin A (1995), l-Deprenyl on behavior and plasma monoamine metabolites in hyperactive adults. Psychopharmacol Bull 31:565Google Scholar
Firestone P, Monteiro-Musten L, Pisterman S, Mercer J, Bennet S (1998), Short-term side effects of stimulant medication are increased in preschool children with attention-deficit hyperactivity disorder. J Child Adolesc Psychopharmacol 8: 13–25Crossref, Google Scholar
Frazier JA, Meyer MC, Biederman J et al. (1999), Risperidone treatment for juvenile bipolar disorder: a retrospective chart review. J Am Acad Child Adolesc Psychiatry 38: 960–965Crossref, Google Scholar
Frierson RL, Wey JJ, Tabler JB (1991), Psychostimulants for depression in the medically ill. Am Fam Phys 43: 163–170Google Scholar
Gadow K, Sverd J, Sprafkin J, Nolan E, Ezor S (1995), Efficacy of methylphenidate for attention deficit hyperactivity in children with tic disorder. Arch Gen Psychiatry 52: 444–455Crossref, Google Scholar
Gillberg C, Melander H, von Knorring A et al. (1997), Long-term central stimulant treatment of children with attention-deficit hyperactivity disorder: a randomized double-blind placebo-controlled trial. Arch Gen Psychiatry 54: 857–864Crossref, Google Scholar
Gittelman-Klein R (1980), Diagnosis and drug treatment of childhood disorders: attention deficit disorder with hyperactivity. In: Diagnosis and Drug Treatment of Psychiatric Disorders: Adults and Children, Klein DF, Gittelman-Klein R, Quitkin F, Rifkin A, eds. Baltimore: Williams & WilkinsGoogle Scholar
Gittelman-Klein R (1987), Pharmacotherapy of childhood hyperactivity: an update. In: Psychopharmacology: The Third Generation of Progress, Meltzer HY, ed. New York: RavenGoogle Scholar
Gittelman-Klein R, Landa B, Mattes JA, Klein DF (1988), Methylphenidate and growth in hyperactive children. Arch Gen Psychiatry 45: 1127–1130Crossref, Google Scholar
Goldman L, Genel M, Bezman R, Slanetz P (1998), Diagnosis and treatment of attention-deficit/hyperactivity disorder. JAMA 279: 1100–1107Crossref, Google Scholar
Grace T (2000), Cellular and molecular neurochemistry of psychostimulant effects on dopamine. In: The Neuropharmacology of Psychostimulant Drugs: Implications for ADHD, Solanto M, Castellanos X, eds. New York: Oxford University Press, pp 85–100Google Scholar
Grade C, Redford B, Chrostowski J, Toussaint L, Blackwell B (1998), Methylphenidate in early poststroke recovery: a double-blind, placebo-controlled study. Arch Phys Med Rehabil 79: 1047–1050Crossref, Google Scholar
Greenhill L (1998a), Childhood attention deficit hyperactivity disorder: pharmacological treatments. In: Treatments That Work, Nathan PE, Gorman J, eds. Philadelphia: Saunders, pp 42–64Google Scholar
Greenhill L (1998b), The use of psychoactive medications in preschoolers: indications, safety and efficacy. Can J Psychiatry 43: 576–581Crossref, Google Scholar
Greenhill L, Abikoff H, Conners CK et al. (1996), Medication treatment strategies in the MTA: relevance to clinicians and researchers. J Am Acad Child Adolesc Psychiatry 35: 444–454Crossref, Google Scholar
Greenhill LL, MTA Cooperative Group (1999), Chronic Stimulant Treatment Effects of Weight Acquisition Rates of ADHD Children. Boca Raton, FL: New Clinical Drug Evaluation Unit Program Conference Proceeding 39: 26–27Google Scholar
Gualtieri CT (1991), Neuropsychiatry and Behavioral Pharmacology. New York: Springer-VerlagGoogle Scholar
Gualtieri CT, Kanoy R, Koriath U et al. (1981), Growth hormone and prolactin secretion in adults and hyperactive children relation to methylphenidate serum levels. Psychoneuroendocrinology 6: 331–339Crossref, Google Scholar
Gualtieri CT, Wargin W, Kanoy R et al. (1982), Clinical studies of methylphenidate serum levels in children and adults. J Am Acad Child Psychiatry 21: 19–26Crossref, Google Scholar
Handen BL, Feldman JM, Lurier A, Murray PJH (1999), Efficacy of methylphenidate among preschool children with developmental difficulties. J Am Acad Child Adolesc Psychiatry 38: 805–812Crossref, Google Scholar
Hinshaw S, Heller T, McHale J (1992), Covert antisocial behavior in boys with attention-deficit hyperactivity disorder: external validation and effects of methylphenidate. J Consult Clin Psychol 60: 274–281Crossref, Google Scholar
Hinshaw S, Henker B, Whalen C, Ehrardy D, Dunnington RE (1989), Aggressive, prosocial and nonsocial behavior in hyperactive boys: dose effects of MPH in naturalistic settings. J Consult Clin Psychol 57: 636–643Crossref, Google Scholar
Jacobvitz D, Srouge LA, Stewart M, Leffert N (1990), Treatment of attentional and hyperactivity problems in children with sympathomimetic drugs: a comprehensive review. J Am Acad Child Adolesc Psychiatry 29: 677–688Crossref, Google Scholar
Jensen P, Kettle L, Roper M et al. (1999), Are stimulants overprescribed? Treatment of ADHD in four US communities. J Am Acad Child Adolesc Psychiatry 38: 797–804Crossref, Google Scholar
Kent J, Blader J, Koplewicz H, Abikoff H, Foley C (1995), Effects of late-afternoon methylphenidate administration on behavior and sleep in attention-deficit hyperactivity disorder. Pediatrics 96: 320–325Google Scholar
Klein R, Abikoff H, Klass E, Ganales D, Seese L, Pollack S (1997), Clinical efficacy of methylphenidate in conduct disorder with and without attention deficit hyperactivity disorder. Arch Gen Psychiatry 54: 1073–1080Crossref, Google Scholar
Kutcher S (1986), Assessing and treating attention deficit disorder in adolescents: the clinical application of a single-case research design. Br J Psychiatry 149: 710–715Crossref, Google Scholar
Lambert NM, Hartsough CS (1998), Prospective study of tobacco smoking and substance dependence among samples of ADHD and non-ADHD subjects. J Learn Disabil 31: 533–544Crossref, Google Scholar
Laufer MW, Denhoff E, Solomon G (1957), Hyperkinetic impulsive disorder in children’s behavior problems. Psychosom Med 19: 38–49Crossref, Google Scholar
Law SF, Schachar R (1999), Do typical clinical doses of methylphenidate cause tics in children treated for attention deficit hyperactivity disorder? J Am Acad Child Adolesc Psychiatry 38:944–951Crossref, Google Scholar
LeFever G, Sawson KV, D, Morrow AL (1999), The extent of drug therapy for attention deficit-hyperactivity disorder among children in public schools. Am J Public Health 89: 1359–1364Crossref, Google Scholar
Loney J, Milich R (1982), Hyperactivity, inattention, and aggression in clinical practice. In: Advances in Developmental and Behavioral Pediatrics, Vol 3, Gadow K, Bialer I, eds. Greenwich, CT: JAI PressGoogle Scholar
Manuzza S, Klein R, Bonagura N, Malloy P, Giampino T, Addlii K (1991), Hyperactive boys almost grown up, V: replication of psychiatric status. Arch Gen Psychiatry 48: 77–83Crossref, Google Scholar
Matochik J, Nordahl T, Gross M et al. (1993), Effects of acute stimulant medication on cerebral metabolism in adults with hyperactivity. Neuropsychopharmacology 8: 377–386Crossref, Google Scholar
Mattes JA, Boswell J, Oliver H (1984), Methylphenidate effects on symptoms of attention deficit disorder in adults. Arch Gen Psychiatry 41: 449–456Crossref, Google Scholar
Mayes S, Crites D, Bixler E, Humphrey B, Mattison R (1994), Methylphenidate and ADHD: influence of age, IQ and neurodevelopmental status. Dev Med Child Neurol 36: 1099–1107Crossref, Google Scholar
McDougle CJ, Holmes JP, Bronson MR et al. (1997), Risperidone treatment of children and adolescents with pervasive developmental disorders: a prospective, open-label study. J Am Acad Child Adolesc Psychiatry 36: 685–693Crossref, Google Scholar
McMaster University Evidence-Based Practice Center (1998), The Treatment of Attention-Deficit/Hyperactivity Disorder: An Evidence Report (Contract 290-97-0017). Washington, DC: Agency for Health Care Policy and ResearchGoogle Scholar
Mitler MM, Hajdukovic R (1990), Narcolepsy. J Clin Neurophysiol 7: 93–118Crossref, Google Scholar
Montieiro-Musten L, Firestone P, Pisterman S, Bennet S, Mercer J (1997), Effects of methylphenidate on preschool children with ADHD: cognitive and behavioral functions. J Am Acad Child Adolesc Psychiatry 36: 1407–1415Crossref, Google Scholar
MTA Cooperative Group (1999a), 14-Month randomized clinical trial of treatment strategies for attention deficit hyperactivity disorder. Arch Gen Psychiatry 56: 1073–1086Crossref, Google Scholar
MTA Cooperative Group (1999b), Moderators and mediators of treatment response for children with ADHD: the MTA study. Arch Gen Psychiatry 56: 1088–1096Crossref, Google Scholar
Murphy DA, Pelham WE, Lang AR (1986), Methylphenidate effects on aggressiveness in ADD and ADD/CD children. Presented at the American Psychological Association, Washington, DCGoogle Scholar
NIH Consensus Statement (1998), Diagnosis and treatment of attention deficit hyperactivity disorder (ADHD). NIH Consens Statement 16: 1–37Google Scholar
Page JG, Bernstein JE, Janicki RS, Michelli FA (1974), A multicenter trial of pemoline (Cylert) in childhood hyperkinesis. In: Clinical Use of Stimulant Drugs in Children, Conners, CK ed. The Hague: Excerpta MedicaGoogle Scholar
Pataki C, Carlson G, Kelly K, Rapport M (1993), Side effects of methylphenidate and desipramine alone and in combination in children. J Am Acad Child Adolesc Psychiatry 32: 1065–1072Crossref, Google Scholar
Patrick KS, Mueller RA, Gualtieri CT, Breese GR (1987), Pharmacokinetics and actions of methylphenidate. In: Psychopharmacology: A Third Generation of Progress, Meltzer HY, ed. New York: Raven, pp 1387–1395Google Scholar
Pelham WE, Burrows-Maclean L, Gnagy EM et al. (2000), Once-a-day Concerta methylphenidate versus t.i.d. methylphenidate in natural settings: safety and efficacy. Pediatrics 121: 126–137Google Scholar
Pelham WE, Greenslade KE, Vodde-Hamilton MA et al. (1990a), Relative efficacy of long-acting stimulants on ADHD children: a comparison of standard methylphenidate, Ritalin-SR, Dexedrine spansule, and pemoline. Pediatrics 86: 226–237Google Scholar
Pelham WE, McBurnett K, Harper G et al. (1990b), Methylphenidate and baseball playing in ADD children: who’s on first? J Consult Clin Psychol 22: 131–135Google Scholar
Pelham WE, Milich R (1991), Individual differences in response to Ritalin in classwork and social behavior. In: Ritalin: Theory and Patient Management, Greenhill LL, Osman B, eds. New York: Mary Ann Liebert, pp 203–222Google Scholar
Pelham WE, Sturges J, Joza Jeal (1989), The effects of sustained release 20 and 10 mg Ritalin b.i.d. on cognitive and social behavior in children with attention deficit disorder. Pediatrics 80: 491–501Google Scholar
Pelham WE, Swanson J, Furman M, Schwindt H (1995), Pemoline effects on children with ADHD: a time-response by dose-response analysis on classroom measures. J Am Acad Child Adolesc Psychiatry 34: 1504–1513Crossref, Google Scholar
Pelham WE, Waschbusch DA (1999), Behavioral interventions in attention deficit/hyperactivity disorder. In: Handbook of Disruptive Behavior Disorders, Quay H, Hogan A, eds. New York: Kluwer Academic/Plenum Publishers, pp 255–278Google Scholar
Perel JM, Greenhill LL, Curran S, Feldman B, Puig-Antich J (1991), Correlates of pharmacokinetics and attentional measures in methylphenidate treated hyperactive children. Clin Pharmacol Ther 49: 160–161Google Scholar
Physicians’ Desk Reference (2000), Montvale, NJ: Medical Economics CompanyGoogle Scholar
Pliszka SR (1992), Comorbidity of attention-deficit hyperactivity disorder and overanxious disorder. J Am Acad Child Adolesc Psychiatry 31: 197–203Crossref, Google Scholar
Pliszka SR (2000), Comparing the effects of stimulant and non-stimulant agents on catecholamine function: implications for theories of attention deficit hyperactivity disorder (ADHD). In: The Neuropharmacology of Psychostimulant Drugs: Implications for AD/HD, Solanto M, Castellanos X, eds. New York: Oxford University Press, pp 141–160Google Scholar
Pliszka SR, Greenhill LL, Crismon ML et al. (2000a), The Texas Children’s Medication Algorithm Project: Report of the Texas Consensus Conference Panel on Medication Treatment of Childhood Attention-Deficit/Hyperactivity Disorder, part I. J Am Acad Child Adolesc Psychiatry 39:908–919Crossref, Google Scholar
Pliszka SR, Greenhill LL, Crismon L et al. (2000b), The Texas Children’s Medication Algorithm Project: Report of the Texas Expert Consensus Conference Panel on Medication Treatment of Childhood Attention-Deficit/Hyperactivity Disorder, part II: tactics. J Am Acad Child Adolesc Psychiatry 39: 920–927Crossref, Google Scholar
Popper CW, Zimnitzky B (1995), Sudden death putatively related to desipramine treatment in youth: a fifth case and a review of speculative mechanisms. J Child Adolesc Psychopharmacol 5: 283–300Crossref, Google Scholar
Rapoport JL, Buchsbaum MS, Weingartner H, Zahn P, Ludlow C, Mikkelsen EJ (1980), Dextroamphetamine: cognitive and behavioral effects in normal and hyperactive boys and normal men. Arch Gen Psychiatry 37: 933–943Crossref, Google Scholar
Rapport M, Carlson G, Kelly K, Pataki C (1993), Methylphenidate and desipramine in hospitalized children, I: separate and combined effects on cognitive function. J Am Acad Child Adolesc Psychiatry 32: 333–342Crossref, Google Scholar
Rapport MD, DuPaul GJ, Kelly KL (1989), Attention deficit hyperactivity disorder and methylphenidate: the relationship between gross body weight and drug response in children. Psychopharmacol Bull 25: 285–290Google Scholar
Ratey J, Greenberg MS, Bemporad JR, Lindem K (1992), Unrecognized attention-deficit hyperactivity disorder in adults presenting for outpatient psychotherapy. J Child Adolesc Psychopharmacol 2: 267–275Crossref, Google Scholar
Richters J, Arnold L, Abikoff H et al. (1995), The National Institute of Mental Health Collaborative Multisite Multimodal Treatment Study of Children With Attention-Deficit Hyperactivity Disorder (MTA), I: background and rationale. J Am Acad Child Adolesc Psychiatry 34: 987–1000Crossref, Google Scholar
Robinson TE, Camp DM (1987), Long-lasting effects of escalating doses of d-amphetamine on brain monoamines, amphetamine-induced stereotyped behavior and spontaneous nocturnal locomotion. Pharmacol Biochem Behav 26: 821–827Crossref, Google Scholar
Rosenberg PB, Ahmed I, Hurwitz S (1991), Methylphenidate in depressed medically ill patients. J Clin Psychiatry 52: 263–267Google Scholar
Sabelsky D (1990), Fluoxetine in adults with residual attention deficit disorder and hypersomnolence. J Neuropsychiatry Clin Neurosci 2: 463–464Crossref, Google Scholar
Safer D, Zito J, Fine E (1996), Increased methylphenidate usage for attention deficit hyperactivity disorder in the 1990s. Pediatrics 98: 1084–1088Google Scholar
Safer DJ, Allen RP (1989), Absence of tolerance to the behavioral effects of methylphenidate in hyperactive and inattentive children. J Pediatr 115: 1003–1008Crossref, Google Scholar
Sallee F, Stiller R, Perel J (1992), Pharmacodynamics of pemoline in attention deficit disorder with hyperactivity. J Am Acad Child Adolesc Psychiatry 31: 244–251Crossref, Google Scholar
Sallee FR, Stiller R, Perel J, Bates T (1985), Oral pemoline kinetics in hyperactive children. Clin Pharmacol Ther 37: 606–609Crossref, Google Scholar
Satterfield JH, Cantwell DP, Satterfield BT (1979), Multimodality treatment: a one-year follow-up of 84 hyperactive boys. Arch Gen Psychiatry 36: 965–974Crossref, Google Scholar
Schachar R, Tannock R (1993), Childhood hyperactivity and psychostimulants: a review of extended treatment studies. J Child Adolesc Psychopharmacol 3: 81–97Crossref, Google Scholar
Schachar R, Tannock R, Cunningham C, Corkum P (1997), Behavioral, situational, and temporal effects of treatment of ADHD with methylphenidate. J Am Acad Child Adolesc Psychiatry 36: 754–763Crossref, Google Scholar
Schleiffer M, Weiss G, Cohen N, Elman M, Cvejic H, Kruger E (1975), Hyperactivity in preschoolers and the effect of methylphenidate. Am J Orthopsychiatry 45: 38–50Crossref, Google Scholar
Schmidt K, Solanto MV, Sanchez-Kappraff M, Vargas P, Wein S (1984), The effect of stimulant medication on academic performance, in the context of multimodal treatment, in attention deficit disorders with hyperactivity: two case reports. J Clin Psychopharmacol 4: 100–103Crossref, Google Scholar
Sebrechts MM, Shaywitz SE, Shaywitz BA, Jatlow P, Anderson GM, Cohen DJ (1986), Components of attention, methylphenidate dosage, and blood levels in children with attention deficit disorder. Pediatrics 77: 222–228Google Scholar
Sherman M (1991), Prescribing practice of methylphenidate: the Suffolk County study. In: Ritalin: Theory and Patient Management, Osman B, Greenhill LL, eds. New York: Mary Ann Liebert, pp 401–420Google Scholar
Shuster L, Hudson J, Anton M, Right D (1982), Sensitization of mice to methylphenidate. Psychopharmacology 71: 31–36Crossref, Google Scholar
Spencer T, Biederman J, Harding M, Faraone S, Wilens T (1996a), Growth deficits in ADHD children revisited: evidence for disorder related growth delays. J Am Acad Child Adolesc Psychiatry 35: 1460–1467Crossref, Google Scholar
Spencer T, Biederman J, Wilens T, Harding M, O’Donnell D, Griffin S (1996b), Pharmacotherapy of attention-deficit hyperactivity disorder across the life cycle. J Am Acad Child Adolesc Psychiatry 35: 409–432Crossref, Google Scholar
Spencer T, Biederman J, Wilens T, Prince J (1995a), Effectiveness and tolerability of tomoxetine in adults with attention-deficit/hyperactivity disorder. In: Proceedings of the Annual Meeting of the American Academy of Child and Adolescent Psychiatry 11:109Google Scholar
Spencer T, Wilens T, Biederman J, Faraone S, Ablon JS, Lapey K (1994), A double-blind comparison of MPH and placebo in adults with attention deficit hyperactivity disorder (ADHD). In: Proceedings of the Annual Meeting of the American Academy of Child and Adolescent Psychiatry 9:42Google Scholar
Spencer T, Wilens T, Biederman J, Faraone S, Ablon JS, Lapey K (1995b), A double-blind, crossover comparison of methylphenidate and placebo in adults with childhood onset ADHD. Arch Gen Psychiatry 52: 434–443Crossref, Google Scholar
Sprague RL, Sleator EK (1977), Methylphenidate in hyperkinetic children: differences in dose effects on learning and social behavior. Science 198: 1274–1276Crossref, Google Scholar
Swanson J (1992), School-Based Assessments and Interventions for ADD Students. Irvine, CA: KC PublishingGoogle Scholar
Swanson J (1993), Effect of stimulant medication on hyperactive children: a review of reviews. Except Child 60: 154–162Google Scholar
Swanson J, Greenhill L, Pelham WE et al. (2000), Initiating Concerta® (OROS methylphenidate HCL) qd in children with attention-deficit/hyperactivity disorder. J Clin Res 3: 59–76Google Scholar
Swanson J, Gupta S, Guinta D et al. (1999a), Acute tolerance to methylphenidate in the treatment of attention deficit hyperactivity disorder in children. Clin Pharmacol Ther 66: 295–305Crossref, Google Scholar
Swanson J, Kinsbourne M, Roberts W, Zucker K (1978), Time-response analysis of the effect of stimulant medication on the learning ability of children referred for hyperactivity. Pediatrics 61: 2–29Google Scholar
Swanson J, Lerner M, Williams L (1995), More frequent diagnosis of attention deficit-hyperactivity disorder. N Engl J Med 333:944Crossref, Google Scholar
Swanson J, Volkow N (2000), Pharmacodynamics and pharmacokinetics of stimulants in AD/HD In: The Neuropharmacology of Psychostimulant Drugs: Implications for AD/HD, Solanto M, Castellanos X, eds. New York: Oxford University Press, pp 101–125Google Scholar
Swanson J, Wigal S, Greenhill L et al. (1998), Analog classroom assessment of Adderall in children with ADHD. J Am Acad Child Adolesc Psychiatry 37: 1–8Crossref, Google Scholar
Swanson JM, Connor DF, Cantwell D (1999b), Combining methylphenidate and clonidine: ill-advised. J Am Acad Child Adolesc Psychiatry 38: 617–619Crossref, Google Scholar
Tannock R, Ickowicz A, Schachar R (1995a), Differential effects of MPH on working memory in ADHD children with and without comorbid anxiety. J Am Acad Child Adolesc Psychiatry 34: 886–896Crossref, Google Scholar
Tannock R, Schachar R, Logan GD (1995b), Methylphenidate and cognitive flexibility: dissociated dose effects in hyperactive children. J Abnorm Child Psychol 23: 235–267Crossref, Google Scholar
Thurber S, Walker C (1983), Medication and hyperactivity: a meta-analysis. J Gen Psychiatry 108: 79–86Google Scholar
Varley C (2000), Sudden death of a child treated with imipramine. J Child Adolesc Psychopharmacol 10: 321–325Crossref, Google Scholar
Varley CK, McClellan J (1997), Case study: two additional sudden deaths with tricyclic antidepressants. J Am Acad Child Adolesc Psychiatry 36: 390–394Crossref, Google Scholar
Vitiello B, Burke L (1998), Generic methylphenidate versus brand Ritalin: which should be used? In: Ritalin: Theory and Practice, Greenhill L, Osman B, eds. Larchmont, NY: Mary Ann Liebert, pp 221–226Google Scholar
Vitiello B, Jensen P (1997), Medication development and testing in children and adolescents. Arch Gen Psychiatry 54: 871–876Crossref, Google Scholar
Volkow N, Ding J, Fowler G et al. (1995), Is methylphenidate like cocaine? Arch Gen Psychiatry 52: 456–464Crossref, Google Scholar
Volkow N, Wang G, Fowler J et al. (1998), Dopamine transporter occupancies in the human brain induced by therapeutic doses of oral methylphenidate. Am J Psychiatry 155: 1325–1331Crossref, Google Scholar
Wallace AE, Kofoed LL, West AN (1995), Double-blind, placebo-controlled trial of methylphenidate in older, depressed medically ill patients. Am J Psychiatry 152: 929–931Crossref, Google Scholar
Weiner N (1991), Drugs that inhibit adrenergic nerves and block adrenergic receptors. In: The Pharmacological Basis of Therapeutics: Norepinephrine, Epinephrine and the Sympathomimetic Amines, Gilman A, Goodman L, eds. New York, pp 145–180Google Scholar
Wender P, Reimherr F (1990), Bupropion treatment of attention-deficit hyperactivity disorder in adults. Am J Psychiatry 147: 1018–1020Crossref, Google Scholar
Wender P, Reimherr F, Wood D (1985), A controlled study of methylphenidate in the treatment of attention deficit disorder. Am J Psychiatry 142: 547–552Crossref, Google Scholar
Wender P, Wood D, Reimherr F, Ward M (1994), An open trial of pargyline in the treatment of adult attention deficit disorder, residual type. Psychiatry Res 9: 329–336Crossref, Google Scholar
Wender PH, Reimher F, Wood D (1981), Attention deficit disorder in adults. Arch Gen Psychiatry 38: 449–456Crossref, Google Scholar
Wigal S, Gupta S, Guinta D, Swanson J (1998), Reliability and validity of the SKAMP Rating Scale in a laboratory school setting. Psychopharmacol Bull 34: 47–53Google Scholar
Wilens T, Biederman J, Spencer T (1994a), Comorbidity of attention-deficit hyperactivity and psychoactive substance use disorders. Hosp Community Psychiatry 45: 421–426Google Scholar
Wilens T, Biederman J, Spencer T (1994b), Clonidine for sleep disturbances associated with attention deficit hyperactivity disorder. J Am Acad Child Adolesc Psychiatry 33: 424–427Crossref, Google Scholar
Wilens T, Biederman J, Spencer T et al. (1999), A pilot controlled trial of ABT-418, a cholinergic agonist, in the treatment of adults with attention-deficit/hyperactivity disorder. Am J Psychiatry 156: 1931–1937Google Scholar
Wilens TE, Biederman J (1992), The stimulants. Psychiatr Clin North Am 15: 191–222Google Scholar
Wilens TE, Spencer TJ (1999), Combining methylphenidate and clonidine: a clinically sound medication option. J Am Acad Child Adolesc Psychiatry 38: 614–616Crossref, Google Scholar
Wolraich M, Greenhill LL, Abikoff H et al. (2001), Randomized controlled trial of OROS methylphenidate once a day in children with attention-deficit/hyperactivity disorder. Pediatrics 108: 883–892Crossref, Google Scholar
Yee JD, Berde CB (1994), Dextroamphetamine or methylphenidate as adjuvants to opioid analgesia for adolescents with cancer. J Pain Symptom Manage 9:122–125Crossref, Google Scholar
Zametkin AJ, Nordahl TE, Gross M et al. (1991), Cerebral glucose metabolism in adults with hyperactivity of childhood onset. N Engl J Med 323: 1361–1366Crossref, Google Scholar
Zametkin AJ, Rapoport JL (1987), Neurobiology of attention deficit disorder with hyperactivity: where have we come in 50 years? J Am Acad Child Adolesc Psychiatry 26:676–686Crossref, Google Scholar
Zito J, Safer D, dosReis S, Gardiner J, Boles M, Lynch F (2000), Trends in the prescribing of psychotropic medications to preschoolers. JAMA 283: 1025–1030Crossref, Google Scholar



