The Role of Behavioral Interventions in Buprenorphine Maintenance Treatment: A Review
Abstract
Objective:
Although counseling is a required part of office-based buprenorphine treatment of opioid use disorders, the nature of what constitutes appropriate counseling is unclear and controversial. The authors review the literature on the role, nature, and intensity of behavioral interventions in office-based buprenorphine treatment.
Method:
The authors conducted a review of randomized controlled studies testing the efficacy of adding a behavioral intervention to buprenorphine maintenance treatment.
Results:
Four key studies showed no benefit from adding a behavioral intervention to buprenorphine plus medical management, and four studies indicated some benefit for specific behavioral interventions, primarily contingency management. The authors examined the findings from the negative trials in the context of six questions: 1) Is buprenorphine that effective? 2) Is medical management that effective? 3) Are behavioral interventions that ineffective in this population? 4) How has research design affected the results of studies of buprenorphine plus behavioral treatment? 5) What do we know about subgroups of patients who do and those who do not seem to benefit from behavioral interventions? 6) What should clinicians aim for in terms of treatment outcome in buprenorphine maintenance?
Conclusions:
High-quality medical management may suffice for some patients, but there are few data regarding the types of individuals for whom medical management is sufficient. Physicians should consider a stepped-care model in which patients may begin with relatively nonintensive treatment, with increased intensity for patients who struggle early in treatment. Finally, with 6-month retention rates seldom exceeding 50% and poor outcomes following dropout, we must explore innovative strategies for enhancing retention in buprenorphine treatment.
(Reprinted with permission from Am J Psychiatry 2017;174:738-747)
The Drug Abuse Treatment Act of 2000 allowed physicians, for the first time, to prescribe an opioid medication, the partial agonist buprenorphine, to treat patients with opioid dependence as part of office-based practice (1). Included in this important act was a proviso that when prescribing buprenorphine in office-based practice, “the practitioner has the capacity to refer the patients for appropriate counseling and other appropriate ancillary services.” Sixteen years later, we address the question, “What is ‘appropriate counseling and other appropriate ancillary services’?”
Four influential randomized trials evaluating the impact of adding counseling or other behavioral interventions to buprenorphine plus medical management have reported no difference in opioid use outcomes between patients receiving and those not receiving additional counseling (2–5). These findings have sparked controversy and confusion among clinicians regarding the role of behavioral interventions in office-based buprenorphine treatment. In this review, after presenting an overview of the four trials, we build on recent reviews (6–8) by synthesizing what the literature reveals about the role of behavioral interventions in buprenorphine treatment as well as identifying important gaps in the literature. We focus the review by considering six key questions raised by the four studies revealing no effect of adding counseling to buprenorphine plus medical management: 1) Is buprenorphine that effective? 2) Is medical management that effective? 3) Are behavioral interventions that ineffective in this population? 4) How has research design affected the results of studies of buprenorphine and behavioral treatment? 5) What do we know about subgroups of patients who do and those who do not seem to benefit from behavioral interventions? 6) What should clinicians aim for in terms of treatment outcome in buprenorphine treatment?
Trials Showing No Benefit of Adding Counseling to Buprenorphine Plus Medical Management
In the first of four studies to date that have found no benefit of adding counseling to office-based buprenorphine plus medical management, Fiellin et al. (5) randomly assigned 166 opioid-dependent patients (83% of whom were primarily heroin users) to one of three treatments: 1) manual-based standard medical management plus weekly dispensing of buprenorphine-naloxone (referred to hereafter as simply “buprenorphine”); 2) standard medical management plus medication dispensing three times a week; and 3) enhanced medical management plus medication dispensing three times a week. All medical management sessions were delivered by primary care nurses who had relatively little experience in treating substance-using individuals. Standard medical management sessions, which lasted approximately 20 minutes, included discussion of recent drug use, support for patients’ attempts to reduce or stop drug use, encouragement of abstinence, and recommendations for mutual-help groups such as Narcotics Anonymous. Enhanced medical management sessions lasted 45 minutes, covering similar topics more deeply. Outcomes improved in all three groups; the self-reported mean number of days per week of opioid use dropped from 5.3 at baseline to 0.4 during treatment. The percentage of opioid-negative urine tests ranged from 40% to 44% among the three treatment arms, with no significant differences. Forty-five percent of patients who underwent randomization completed the 24-week trial.
More recently, Fiellin et al. (4) examined the effect of adding cognitive-behavioral therapy (CBT) to buprenorphine plus standard medical management in a 24-week study of 141 opioid-dependent primary care patients (64% of them primarily heroin users). Physicians delivered medical management weekly for 2 weeks, then biweekly for 4 weeks, then every 4 weeks. Patients who were randomly assigned to receive medical management plus CBT were offered up to 12 CBT sessions in weeks 1–12. Again, there were significant improvements in frequency of self-reported opioid use, with a reduction from a mean of 5.3 days of opioid use per week at baseline to a mean of 0.4 days per week during the second half of buprenorphine maintenance, with no significant difference between study conditions. Forty-one percent of participants who underwent randomization completed the trial.
Ling et al. (2) tested CBT, contingency management, and their combination against medical management alone in a 32-week trial of 202 opioid-dependent patients (59% of them primarily heroin users). All participants received weekly medical management, and they were randomly assigned to receive weekly CBT, twice-weekly contingency management (whereby patients with drug-free urine tests received a chance to receive cash rewards ranging from $1 to $4), contingency management plus CBT, or medical management alone. Participants in all four conditions reported significant improvements in opioid use: days of heroin use in the past 30 days dropped from >20 in all conditions to 3.3–5.4 at the end of behavioral treatment, with no significant differences among groups. Fifty percent of participants who underwent randomization completed the trial.
The largest study to examine the role of counseling in office-based buprenorphine treatment was the Prescription Opioid Addiction Treatment Study (POATS) (3), a 10-site trial (N=653) conducted by the National Drug Abuse Treatment Clinical Trials Network. This study focused on individuals dependent on prescription opioids, either exclusively (77%) or with a history of minimal, non-injection heroin use (23%). Using an adaptive treatment research design, POATS randomly assigned patients to weekly medical management or medical management plus individual drug counseling during 1) a 4-week taper and 2) for those who relapsed during the first phase, 12 weeks of buprenorphine stabilization. The rate of treatment success (abstinence or near-abstinence from opioids) was only 7% during the taper phase, but it rose to 49% in the second phase of the trial (N=360) during the last 4 weeks of buprenorphine stabilization. Again, there was no significant difference in success rates between those who received counseling and those who did not. After completing phase 2, participants were tapered off buprenorphine; only 9% of patients were abstinent after completing a second taper and 8 weeks of follow-up, again underscoring the importance of maintaining patients on buprenorphine for longer periods. The second phase of the trial included 360 of the 610 participants (59%) who did not succeed in phase 1; 90% of phase 2 participants completed the 12 weeks of buprenorphine stabilization treatment.
In light of the similar results from these four trials, which indicate improvement in opioid use with buprenorphine plus medical management but no benefit from additional behavioral interventions, we explore potential explanations below.
Buprenorphine Treatment and the Role of Behavioral Interventions: Six Questions
Is Buprenorphine That Effective?
Are the effects of buprenorphine so powerful as to overwhelm those of behavioral therapies? As is true with most pharmacotherapies for substance use disorders, one’s view of the effectiveness of buprenorphine depends on the question, “Compared to what?” There is no doubt that office-based buprenorphine treatment constitutes a significant advance in broadening the availability of an effective treatment for opioid dependence and has saved many lives (9–12). Numerous reviews and meta-analyses underscore the effectiveness of buprenorphine in enhancing treatment retention and reducing illicit opioid use compared with placebo or no treatment (13, 14). Buprenorphine’s important benefits also include reduced risk of HIV and sexually transmitted diseases, reduced medical costs, and reduced mortality (9).
However, there is clear room for improvement in buprenorphine treatment outcomes. In the POATS study (3), the sevenfold increase in rates of successful outcome for buprenorphine stabilization compared with a taper is a clear testament to its effectiveness; the fact that half of the participants were unsuccessful on buprenorphine stabilization highlights its limitations. Indeed, when compared with methadone maintenance, buprenorphine consistently demonstrates significantly lower rates of retention (13–17). For example, in a multisite clinical trial of 1,267 individuals randomly assigned to receive methadone or buprenorphine maintenance, the 6-month retention rates for buprenorphine and methadone were 46% and 74%, respectively (p<0.01), with most dropout from buprenorphine occurring during the first month (18). A 5-year follow-up study indicated continued superiority for methadone compared with buprenorphine in both retention and rates of illicit opioid use (19).
Although it is possible that the superior retention with methadone over buprenorphine in this randomized trial reflects individuals not being assigned to their preferred treatment, given that opioid-dependent individuals often have strong preferences for one over the other (20), a study by Pinto et al. (21) suggests an alternative explanation. Pinto et al. conducted a practical trial in the United Kingdom in which 361 opioid-dependent individuals were allowed to select whether they would receive buprenorphine or methadone maintenance treatment. The sample included predominantly nonminority unemployed heroin users with an average of 12 years of opioid dependence and substantial treatment histories. Most (63%) chose methadone over buprenorphine. Individuals who selected methadone tended to have more severe substance use problems and more psychological difficulties. Even with self-selection of treatment, 6-month retention rates were markedly higher with methadone compared with buprenorphine (69.6% and 42.5%, respectively; p<0.001). Conversely, individuals who were retained in buprenorphine treatment had significantly fewer opioid-positive urine specimens. Similar findings have been reported in large national data linkage studies (16). It should be noted that in the United Kingdom, both buprenorphine and methadone can be prescribed by physicians in office practice, whereas comparisons of buprenorphine and methadone in the United States involve differences not only in pharmacology (partial versus full agonist) but in delivery (buprenorphine treatment is most often office based, whereas methadone is only available through specialty opioid treatment programs).
Why might methadone be more effective than buprenorphine in retaining patients? In addition to pharmacologic differences, one factor in the United States may be the higher level of structure associated with methadone programs; most provide more regular contact, urine monitoring, and a larger array of counseling and services than office-based buprenorphine treatment (22). The substantial literature on outcomes for methadone maintenance programs indicates large differences in retention and outcome that vary with the intensity and quality of services provided (23). For example, in the landmark study on the effects of behavioral interventions and services in methadone maintenance treatment, McLellan et al. (24) randomly assigned 92 male opioid-dependent individuals who were stabilized on a therapeutic dosage of methadone to receive 1) no additional services, 2) standard services, including counseling, or 3) enhanced services, which provided counseling plus individually tailored on-site medical, psychiatric, employment, and family therapy services. Although some individuals did well in the no-services condition, 69% had to be protectively transferred out of the study because of “unremitting use of opiates or cocaine, or medical/psychiatric emergencies.” Outcomes were significantly different across conditions, with the best outcomes in the enhanced condition and the poorest results in the no-services condition. The authors concluded, “The addition of basic counseling was associated with major increases in efficacy.” That study and many more have led to the general, though not universal (25–27), consensus that behavioral intervention is a key component of successful methadone treatment programs (28). Although the McLellan et al. study has not yet been replicated in the context of buprenorphine maintenance treatment, it raises the question, “Why would the lessons learned from methadone treatment not apply to buprenorphine?” One answer is the difference between delivery of the two medications in the United States; since buprenorphine can be prescribed in a physician’s office, medical management should be, by definition, part of the process of buprenorphine treatment, whereas there is no analogue in methadone treatment. Therefore, medical management itself could represent an effective behavioral treatment that obviates the need for further counseling for some patients.
Is Medical Management That Effective?
The four studies suggesting no additional benefit of behavioral intervention with buprenorphine maintenance treatment all included a crucial component: regular medical management. In those studies, medical management typically involved regular appointments with a physician who assessed participant progress and medication efficacy and side effects and consistently stressed simple messages, including the importance of abstinence and the benefits of attending mutual-help support groups, as well as regular (at least weekly) urine toxicology screens. It is impossible to determine how much the medical management contributed to patient outcomes in those studies; we are aware of no studies evaluating whether behavioral interventions might improve treatment outcomes when buprenorphine is delivered with minimal or no medical management. It is possible that the effect of regular, structured physician-delivered medical management plus urine monitoring was roughly parallel to the “standard methadone services” provided in the McLellan et al. study described above (24), highlighting the potentially powerful influence of physician involvement on patient outcomes.
It is also likely that the medical management delivered in the four studies described above was offered more frequently and in a more structured way than is typical in many community settings (29, 30). It is unclear whether such frequent physician contact and urine monitoring is even feasible in most office-based settings (31, 32). Again, although there have been no studies comparing the effects of different intensities or types of medical management on buprenorphine outcomes, the variable retention rates and outcomes reported among community-based buprenorphine treatment programs suggests that the quality and intensity of medical management varies widely as well (33–37). Hence, in settings where medical management is not provided as intensively as it was in the four studies reviewed, additional behavioral intervention may have a greater effect on outcomes.
Are Behavioral Interventions Ineffective in this Population?
The four cited negative studies (2–5) are countered by four other studies that do point to the efficacy of certain behavioral interventions in buprenorphine maintenance treatment (Table 1). Bickel et al. (38) randomly assigned 135 opioid-dependent individuals to one of three conditions in conjunction with buprenorphine plus thrice-weekly urine monitoring: 1) standard buprenorphine maintenance treatment, which included brief weekly counseling similar to that offered in methadone maintenance settings; 2) the clinician-delivered community reinforcement approach, a validated multicomponent approach (42) that included voucher-based contingency management for providing cocaine- and opioid-free urine toxicology screens (maximum possible earnings for complete abstinence, $1,316); and 3) the community reinforcement approach, provided via computer through the Therapeutic Education System, which also included the same voucher-based contingency management procedure. The rates of retention through 23 weeks were 58%, 53%, and 62%, respectively, and not significantly different by condition. However, participants assigned to the two community reinforcement approach conditions had significantly longer periods of abstinence from both opioids and cocaine and more drug-negative urine specimens (70% for computer-delivered, 73% for therapist-delivered) compared with participants receiving standard counseling (57%).
| Study | N | Conditions | Exclusionsb | Length of Trial | Frequency of Medical Management | Behavioral Therapy | Frequency of Urine Monitoring | 6-Month Retention Ratec | Opioid-Free Urine Specimens |
|---|---|---|---|---|---|---|---|---|---|
| Fiellin et al. 2006 (5) | 166 | 3 | No alcohol, benzodiazepine, or sedative dependence | 24 weeks | 1–3 times a week | Standard medical management, 20 minutes; enhanced management, 45 minutes | Once a week | MM once a week, 50%; MM three times a week, 41%; EMM three times a week, 44% (completion confounded with attendance at counseling) | MM once a week, 44%; MM three times a week, 40%; EMM three times a week, 40% |
| Weiss et al. 2011 (3) | 653 | 2 | No heroin injection or history of heroin dependence or pain event within 6 months; no alcohol or other drug dependence requiring immediate medical attention | 24 weeks (phase 1: 4-week taper, 8-week follow-up; phase 2: 12-week maintenance + 4-week taper + 8-week follow-up) | Phase 1: twice in week 1, then once each in weeks 2, 3, 4, 6, 8. Phase 2: twice a week in week 1, then once a week in weeks 2–16 | Individual manualized drug counseling. Phase 1: twice a week in weeks 1–4, every 2 weeks in weeks 5–8. Phase 2: twice a week in weeks 1–6, once a week in weeks 7–12, no visits in weeks 13–16 | Once a week in weeks 1–16, twice a month in weeks 17–24 | Successful outcome: composite of retention and abstinence. End of phase 1: MM, 7%; MM+ODC, 6%. End of phase 2: MM, 47%; MM+ODC, 52% | Not reported by group; 61% during weeks 1–12; 42% during weeks 13–24 |
| Ling et al. 2013 (2) | 202 | 4 | No alcohol or other drug dependence requiring immediate medical attention | 32 weeks (phase 1: 16 weeks of behavioral treatment; phase 2: 16 weeks with medication maintenance only) | Twice a week for 16 weeks; checklist used | CM twice a week; CBT once a week in weeks 1–16; no behavioral therapy in weeks 17–32 | Twice a week in weeks 1–16, once a week in weeks 17–24 | CBT, 49%; CM, 57%; CBT+CM, 49%; NT, 43% (32-week retention) | CBT, 54%; CM, 46%; CBT+CM, 42%; NT, 52% |
| Fiellin et al. 2013 (4) | 141 | 2 | No alcohol, cocaine, or benzodiazepine dependence | 24 weeks (12 weeks with CBT, 12 weeks no CBT) | Once a week for the first 2 weeks, then twice a month | Clinician-delivered CBT once a week | Once a week | MM , 45%; MM+CBT, 38%. Transfer to methadone: MM, 39%; MM+CBT, 27% | Not reported |
| Bickel et al. 2008 (38) | 135 | 3 | None | 23 weeks | Not described | Standard counseling: once a week; computer CRA: three times a week; clinician CRA: three times a week | Three times a week | Standard counseling, 58%; computer CRA, 62%; clinician CRA, 53% | Standard counseling, 57%; computer CRA, 70%; clinician CRA, 73% |
| Christensen et al. 2014 (39) | 170 | 2 | None | 12 weeks | Not described | Computer CRA three times a week; all received CRA three times a week and 30 minutes of counseling every 2 weeks | Three times a week | CRA+CM, 80%; CM only, 64% (12-week retention) | Not reported |
| Schottenfeld et al. 2005 (40) | 162 | 4 | No alcohol or sedative dependence | 24 weeks; contingencies change at week 12 | Not described | Counseling: twice a week in weeks 1–12, once a week in weeks 13–24 | Three times a week | MMT+CM, 60%; MMT+PF, 75%; BUP+CM, 45%; BUP+PF, 22% | MMT+CM, 55%; MMT+PF, 50%; BUP+CM, 37%; BUP+PF, 28% |
| Miotto et al. 2012 (41) | 94 | 3 | No benzodiazepine or other substance dependence | 20-week assessment; 52 weeks total | Flexible | OTP: once a week in weeks 1–6, once a month in weeks 7–52. Primary care: once a week in weeks 1–6, once a month in weeks 7–52. Matrix Model: weekly groups | Once a week in weeks 1–9, once a month through week 52 | 20-week retention. OTP, 21%; primary care, 33%; Matrix Model, 52% | At week 20: OTP, 22%; primary care, 17%; Matrix Model, 33% |
TABLE 1. Overview of Eight Studies Evaluating Behavioral Interventions and Buprenorphine Maintenancea
Christensen et al. (39) randomly assigned 170 opioid-dependent individuals to receive either buprenorphine with contingency management alone (maximum voucher value, $997) or contingency management combined with the computerized Therapeutic Education System. The combined computerized therapeutic education system plus contingency management was associated with significantly better retention than contingency management alone through the 12-week trial (80% compared with 64%) as well as significantly longer periods of continuous abstinence (a mean of 55 days compared with 50 days). Longer-term follow-up data were not reported for this study.
Schottenfeld et al. (40) randomly assigned 162 cocaine- and opioid-dependent individuals to receive therapeutic dosages of either methadone or buprenorphine and either voucher-based contingency management or performance feedback in a factorial (2×2) design. All participants received weekly individual counseling. In the contingency management system, the value of the vouchers escalated between weeks 1 and 12, then was reduced to a nominal value for weeks 13–24; the maximum possible total value was $1,033. Over the course of the 24-week study, retention was significantly higher for participants assigned to methadone compared with those assigned to buprenorphine (p<0.05); similarly, those assigned to methadone had significantly longer periods of abstinence and submitted significantly more drug-free urine specimens (36% compared with 19%; p<0.05). The overall study retention rate through 24 weeks was 55%. There was a significant effect of contingency management on submission of drug-free urine specimens during the first 12 weeks, the period during which the voucher values escalated (29% compared with 21%; p<0.05), although there were no significant effects for contingency management on retention or submission of drug-free urine specimens over the full 24 weeks. This study illustrates two key points. First, as in the larger body of literature on contingency management interventions, effects of contingency management procedures are most pronounced when escalating contingencies are in effect and tend to weaken once the contingencies are terminated (43). Second, the lower retention associated with buprenorphine may make it more difficult to detect effects of behavioral interventions due to “floor” effects associated with poorer retention, as illustrated in Figure 1.
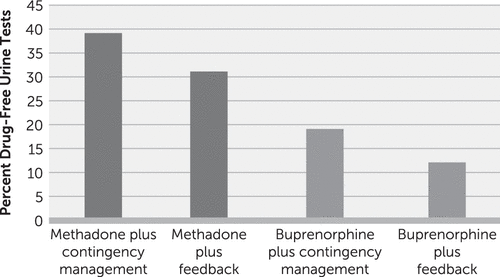
FIGURE 1. Outcomes by Treatment Condition in a 2005 Study of 162 Cocaine- and Opioid-Dependent Individuals Who Received Either Methadone or Buprenorphine Plus Either Contingency Management or Performance Feedbacka
a Data from Schottenfeld et al. (40).
In addition to these three studies showing beneficial effects of behavioral interventions, one study demonstrated mixed results, with positive effects on retention but not on urine toxicology tests. Miotto et al. (41) randomly assigned 94 opioid-dependent individuals to one of three treatment sites in which they received buprenorphine maintenance treatment over a 52-week period: 1) an opioid treatment program that provided supportive counseling by a certified counselor at the time of the medication visit (weekly during weeks 1–6 and monthly during weeks 7–52); 2) a primary care setting in which a physician provided brief counseling at the time of the study visits (also weekly during weeks 1–6 and monthly during weeks 7–52); and 3) behaviorally oriented psychosocial treatment using the Matrix Model, in which CBT groups were offered weekly for weeks 1–52. At week 20, there were significant between-site differences in retention (21% in the opioid treatment program, 33% in the primary care setting, and 51% in the Matrix Model; p=0.05). However, there were no significant differences by site in the rate of opioid-positive urine tests. It should be noted that there were differences in average dosage of buprenorphine prescribed across settings, with physicians prescribing lower dosages in the primary care setting.
Has Research Design Affected the Results of Studies of Buprenorphine and Behavioral Treatment?
As described above, four studies have indicated no additional benefit of adding a behavioral intervention to buprenorphine maintenance treatment, while four others have shown a potential benefit. Is it possible that the four negative studies were designed in a way that influenced the likelihood of finding a benefit of behavioral interventions? These were all rigorous randomized trials with multiple strengths, including comparatively large sample sizes, manualized interventions, and biological indicators of drug use outcomes (regular urine toxicology tests). However, when we compare the designs of the positive and negative trials, two key features emerge that may have influenced the outcomes: the intensity and quality of medical management (both in terms of buprenorphine dosage and medically oriented counseling) and the nature of the behavioral intervention itself (counseling of some sort versus contingency management). In particular, in all four negative studies (2–5), the behavioral interventions tested (counseling, CBT, or contingency management) were provided in addition to comparatively intensive medical management; three of the four studies included weekly medical management meetings. In contrast, three of the four positive studies did not describe a structured medical management condition.
The four negative studies also allowed for higher buprenorphine dosing than did the positive studies; the two Fiellin et al. studies (4, 5) and the Ling et al. study (2) allowed dosages of up to 24 mg/day, while POATS (3) allowed dosages of up to 32 mg/day, with dosage adjustments occurring as part of medical management visits. The Christensen et al. (39) and Schottenfeld et al. studies (40) had maximum daily doses of 16 mg, while the Bickel et al. study (38) had a maximum daily dose of 18 mg unless the participant was in opioid withdrawal. Because there is some evidence that higher dosages of buprenorphine are associated with better opioid use outcomes (15, 18), it is possible that some patients in the four negative studies benefited from their medical management sessions and more liberal dosage adjustments, whereas the behavioral interventions in the positive studies played a more important role.
The nature of the behavioral interventions themselves may have significantly influenced outcome. For example, in the first Fiellin et al. study (5), both the standard and enhanced medical management sessions were delivered by primary care nurses and involved a difference of only 25 minutes per session. Although the second Fiellin et al. study (4) included a somewhat less intensive medical management condition, CBT was offered only once a week, and participants completed an average of only 6.7 CBT sessions over the 12-week treatment period; the Ling et al. study (2) also offered CBT only once a week. The drug counseling offered in POATS (3) was somewhat more intensive (twice weekly in phase 1, and in phase 2, twice weekly for 6 weeks, then weekly for 6 weeks); however, participants attended only 64.4% of potentially available counseling sessions in phase 2, when the main outcome was measured. Thus, in all four of these negative trials, the difference in treatment intensity between the medical management condition and the medical management plus behavioral treatment condition may not have been robust enough to make a difference in outcome. Moreover, all of the studies showing benefit from a behavioral intervention on urine test–confirmed opioid use outcomes entailed contingency management, in the context of either computerized or in-person counseling. Conversely, three of the four negative studies included counseling but no contingency management condition. The Ling et al. study (2) was the only one in which contingency management offered no advantage; however, unlike the three positive contingency management studies, this study included weekly medical management visits, thus possibly mitigating the effect of other interventions. In addition, as effect sizes of behavioral interventions may be modest, it is not clear whether any of the studies other than the multisite POATS were adequately powered to detect a significant difference between the behavioral conditions.
Finally, the handling of missing data and the timing of the measurement of primary outcomes may be important factors in interpreting outcomes from the negative trials. For almost all of the trials reviewed here, dropout rates were high, and data were not available from the roughly 50% of participants who were withdrawn or dropped out. Missing urine screens were assumed to be positive for drug use, and dropouts were assumed to be treatment “failures”; while this assumption remains a common approach in this emerging field, it should be noted that it is highly problematic from a missing data perspective (44) and subject to multiple biases. Multiple studies have demonstrated that it is possible to achieve high rates of follow-up in studies of opioid-dependent patients, and these studies demonstrate that outcomes vary widely across patients who drop out of a study, and include inpatient treatment, switch to a methadone maintenance program, and others (45, 46); thus, the validity of the “missing equals failure” assumption is questionable.
What Do We Know About Subgroups of Patients Who Do and Do Not Benefit From Behavioral Interventions?
One potentially useful way to frame this question may be “what types of patients respond well to medical management alone, in contrast to those patients who may require more intensive behavioral interventions?” To date, very few studies have addressed this issue. A secondary analysis of the POATS study (47) focused on subgroups of participants who benefited from counseling in addition to buprenorphine and medical management, either because of greater problem severity or more exposure to counseling as a result of greater treatment adherence. Problem severity was measured alternately by a history of heroin use, a higher Addiction Severity Index (ASI) drug composite score (48), and presence of current chronic pain. Adequate treatment adherence was defined a priori as attending at least 60% of all offered sessions. The association between severity and outcome did not vary by treatment condition for chronic pain or ASI drug severity composite score. However, among patients with a history of heroin use, those who received drug counseling were more likely to be successful (i.e., abstinent or nearly abstinent from opioids) than those who received medical management alone, but only if they were adherent to treatment and thus received adequate exposure to counseling (p=0.03). This is notable because not only did heroin users as a group have worse overall outcomes in both the POATS main trial and in a 42-month follow-up study of the POATS population (45), but also heroin users have repeatedly demonstrated worse treatment outcomes compared with those who exclusively use prescription opioids (5, 49, 50). These findings emphasize the importance of treatment adherence and suggest that patients with prescription opioid dependence are a heterogeneous group and that optimal treatment strategies may vary by subgroup (47).
A secondary analysis (51) of the 2013 Fiellin et al. study (4) suggested that individuals who were dependent on prescription opioids had a better response to CBT, as measured by percentage of urine tests negative for all drugs (p=0.02), with no significant differences between CBT and medical management for participants who were primary heroin users. We know of no other study that has reported on predictors of response to medical management alone compared with additional behavioral interventions. However, the general literature on outcomes from buprenorphine-maintained patients in office-based settings suggests that better outcomes are associated with attendance at counseling (36, 52–55) or Alcoholics Anonymous or Narcotics Anonymous meetings (36, 54). For example, in a retrospective chart review study of a heterogeneous sample of 533 opioid-dependent individuals enrolled in a primary care center, Neumann et al. (33) reported that retention in treatment through 6 months was associated with attendance at counseling and past physical trauma or injury.
What Outcomes Should Clinicians Aim for in Buprenorphine Maintenance Treatment?
The literature on buprenorphine maintenance treatment in office-based settings overwhelmingly emphasizes two outcomes: treatment retention and submission of urine toxicology tests negative for opioids and other drugs. While these are essential outcome indicators, they do not address other important areas of high significance to patients and their families (56, 57). There are no reliable data, for example, on the extent to which any of the treatment approaches reviewed here are successful in helping patients attain a sustained period of good functioning (which could be defined as having a stable place to live, a job, no criminal activity, and management or resolution of other medical or psychiatric problems). Trials should begin routinely assessing functional outcomes among buprenorphine-maintained patients (as problems in the area of housing, employment, illegal activity, and medical and psychiatric functioning are likely to be associated with poorer buprenorphine outcomes) and consider treatment models in which achieving good functional outcomes is a goal.
Furthermore, as provision of intensive behavioral intervention may be neither necessary for all buprenorphine-treated patients nor feasible in many office-based settings, until we understand more about the subtypes of patients for whom medical management is adequate and those who require more intensive intervention, stepped-care models (58) should be considered and evaluated systematically. Some patients with opioid use disorders do not want or accept counseling (59–61). Indeed, some behavioral interventions for this patient population have involved increased intensity of counseling as a negative consequence of continued substance use (62). A potential stepped-care model could involve starting those patients appropriate for office-based buprenorphine treatment at a level acceptable to them, which might be a basic level of medical management or medical management plus counseling, along with regular urine testing and monitoring; the treatment would consistently focus on what we call the “five A’s” of successful buprenorphine treatment: adherence, abstinence, attendance, alternative activities, and accessing support. For those patients who do well with this model and have adequate support, additional intervention may be needed episodically, during periods of stress, or not at all. However, patients who do poorly at any given level of treatment intensity should be offered a more intensive behavioral intervention. A secondary analysis from POATS (63) revealed that patients who used opioids during the first 2 weeks of buprenorphine treatment had only a 6% likelihood of achieving abstinence at 12 weeks, which is consistent with other research (52). Thus, the physician can tell early in treatment whether the level of behavioral intervention is adequate or needs strengthening.
For those individuals who show signs of needing additional intervention (missed visits, drug-positive urine tests, symptoms that cannot be managed in the office setting), more intensive intervention is indicated. A significant problem is that access to high-quality evidence-based interventions is limited in many areas of the country (11, 64–66). Alternative services and support, such as Alcoholics Anonymous and Narcotics Anonymous, may be more readily available. However, there is virtually no information on how many buprenorphine-treated patients use such services or what benefits they may provide, particularly because there may be substantial variation in how buprenorphine-treated patients are received in some mutual-help groups (66, 67).
Summary and Conclusions
To summarize in terms of the questions raised at the beginning of this review, first, is buprenorphine that effective? While there is no question that buprenorphine maintenance is far more effective than treatments that do not involve approved medications for opioid use disorders, retention in buprenorphine treatment appears to be significantly poorer than in methadone maintenance treatment. Thus, while efforts to expand buprenorphine access are essential and urgent, there remains considerable room for improvement, given 6-month retention rates of about 50% and the significantly higher risk of relapse, overdose, and death that is associated with dropout (16, 68, 69). Given these risks, we must find means of improving retention in office-based buprenorphine maintenance.
Second, is medical management that effective? Our review suggests that medical management, at the levels provided in the four negative studies reviewed, may reflect an adequate level of treatment for many patients. However, it is unclear whether the levels of intensity of medical management provided in these studies are routinely offered or even feasible in most community-based settings. Thus, efforts to identify subtypes of patients who respond to medical management and those who require more intensive care should receive greater attention, in both completed and future studies.
Third, regarding the question of whether behavioral interventions are ineffective in this population, the four studies finding no benefit from behavioral interventions are countered by an equal number of trials demonstrating the efficacy of behavioral interventions, particularly contingency management. This underscores the idea that interventions with a stronger evidence base, such as contingency management, may have an important role in buprenorphine maintenance treatment, and it suggests that the issue regarding the role of behavioral interventions is far from closed.
Fourth, in terms of the extent to which research design may have affected the results of studies of buprenorphine and behavioral treatment, two factors are notable. First, no study has compared a behavioral therapy to buprenorphine maintenance delivered without high-quality medical management. Second, the high dropout rate in these studies presents a general problem with this literature, related to the handling of significant levels of missing data. The assumption that missing data represent categorical treatment failure can open these studies to multiple sources of bias and limit the strength of inferences that can be drawn from them (44, 70). Future studies should use proven methods to achieve high levels of follow-up (71–73), particularly of participants who drop out of treatment.
Finally, in terms of understanding 1) subgroups of patients who do and do not benefit from behavioral interventions, and 2) desirable outcomes from buprenorphine treatment, more data are needed on key issues, including 1) the types of individuals for whom medical management with buprenorphine is adequate, in contrast with those who need more intensive care; 2) rates of buprenorphine-maintained patients who attend mutual-help groups and their positive and negative experiences with these important support groups; 3) outcome studies comparing stepped and fixed models of care; 4) innovative approaches that both appeal to patients on buprenorphine maintenance treatment and improve outcomes beyond the current mark of 50% retention at 6 months; 5) the relationship between buprenorphine dosage and patient outcome; and 6) optimal models of treatment to produce improvements in overall functioning for patients with opioid use disorders.
1 : Clinical Guidelines for the Use of Buprenorphine in the Treatment of Opioid Addiction, TIP Series 40. Rockville, Md, Substance Abuse and Mental Health Services Administration, 2004Google Scholar
2 : Comparison of behavioral treatment conditions in buprenorphine maintenance. Addiction 2013; 108:1788–1798Crossref, Google Scholar
3 : Adjunctive counseling during brief and extended buprenorphine-naloxone treatment for prescription opioid dependence: a 2-phase randomized controlled trial. Arch Gen Psychiatry 2011; 68:1238–1246Crossref, Google Scholar
4 : A randomized trial of cognitive behavioral therapy in primary care-based buprenorphine. Am J Med 2013; 126:74e11–74e17Crossref, Google Scholar
5 : Counseling plus buprenorphine-naloxone maintenance therapy for opioid dependence. N Engl J Med 2006; 355:365–374Crossref, Google Scholar
6 : Prescription opioid misuse, abuse, and treatment in the United States: an update. Am J Psychiatry 2016; 173:18–26Crossref, Google Scholar
7 : A systematic review on the use of psychosocial interventions in conjunction with medications for the treatment of opioid addiction. J Addict Med 2016; 10:93–103Crossref, Google Scholar
8 : Behavioral counseling content for optimizing the use of buprenorphine for treatment of opioid dependence in community-based settings: a review of the empirical evidence. Am J Drug Alcohol Abuse 2007; 33:643–654Crossref, Google Scholar
9 : Buprenorphine in the treatment of opioid addiction: opportunities, challenges, and strategies. Expert Opin Pharmacother 2014; 15:2263–2275Crossref, Google Scholar
10 : Office-based buprenorphine treatment for opioid-dependent patients. Harv Rev Psychiatry 2004; 12:321–338Crossref, Google Scholar
11 : National and state treatment need and capacity for opioid agonist medication-assisted treatment. Am J Public Health 2015; 105:e55–e63Crossref, Google Scholar
12 : A call for evidence-based medical treatment of opioid dependence in the United States and Canada. Health Aff (Millwood) 2013; 32:1462–1469Crossref, Google Scholar
13 : Medication-assisted treatment with buprenorphine: assessing the evidence. Psychiatr Serv 2014; 65:158–170Link, Google Scholar
14 : Methadone and buprenorphine for the management of opioid dependence: a systematic review and economic evaluation. Health Technol Assess 2007; 11:1–171Crossref, Google Scholar
15 : Buprenorphine maintenance versus placebo or methadone maintenance for opioid dependence. Cochrane Database Syst Rev 2014; 2:CD002207Google Scholar
16 : Comparing retention in treatment and mortality in people after initial entry to methadone and buprenorphine treatment. Addiction 2009; 104:1193–1200Crossref, Google Scholar
17 : A comparison of characteristics and outcomes of opioid-dependent patients initiating office-based buprenorphine or methadone maintenance treatment. Subst Abus 2014; 35:122–126Crossref, Google Scholar
18 : Treatment retention among patients randomized to buprenorphine/naloxone compared to methadone in a multi-site trial. Addiction 2014; 109:79–87Crossref, Google Scholar
19 : Long-term outcomes after randomization to buprenorphine/naloxone versus methadone in a multi-site trial. Addiction 2016; 111:695–705Crossref, Google Scholar
20 : Patient perspectives on choosing buprenorphine over methadone in an urban, equal-access system. Am J Addict 2013; 22:285–291Crossref, Google Scholar
21 : The SUMMIT trial: a field comparison of buprenorphine versus methadone maintenance treatment. J Subst Abuse Treat 2010; 39:340–352Crossref, Google Scholar
22 : Office-based maintenance treatment of opioid dependence: how does it compare with traditional approaches? CNS Drugs 2008; 22:99–111Crossref, Google Scholar
23 : The Effectiveness of Methadone Maintenance Treatment. New York, Springer-Verlag, 1991Crossref, Google Scholar
24 : The effects of psychosocial services in substance abuse treatment. JAMA 1993; 269:1953–1959Crossref, Google Scholar
25 : A randomized controlled trial of interim methadone maintenance. Arch Gen Psychiatry 2006; 63:102–109Crossref, Google Scholar
26 : Randomized trial of standard methadone treatment compared to initiating methadone without counseling: 12-month findings. Addiction 2012; 107:943–952Crossref, Google Scholar
27 : A randomized trial of 6-month methadone maintenance with standard or minimal counseling versus 21-day methadone detoxification. Drug Alcohol Depend 2008; 94:199–206Crossref, Google Scholar
28 : Principles of Drug Addiction Treatment: A Research Based Guide. Bethesda, Md, NIDA, 2007Google Scholar
29 : Expanding treatment capacity for opioid dependence with office-based treatment with buprenorphine: national surveys of physicians. J Subst Abuse Treat 2010; 39:96–104Crossref, Google Scholar
30 : Two-year experience with buprenorphine-naloxone (Suboxone) for maintenance treatment of opioid dependence within a private practice setting. J Addict Med 2007; 1:104–110Crossref, Google Scholar
31 : Outcomes of buprenorphine maintenance in office-based practice. J Addict Dis 2007; 26:13–23Crossref, Google Scholar
32 : Office-based management of opioid dependence with buprenorphine: clinical practices and barriers. J Gen Intern Med 2008; 23:1393–1398Crossref, Google Scholar
33 : Primary care patient characteristics associated with completion of 6-month buprenorphine treatment. Addict Behav 2013; 38:2724–2728Crossref, Google Scholar
34 : Collaborative care of opioid-addicted patients in primary care using buprenorphine: five-year experience. Arch Intern Med 2011; 171:425–431Crossref, Google Scholar
35 : Feasibility of implementing shared medical appointments (SMAs) for office-based opioid treatment with buprenorphine: a pilot study. Subst Abus 2015; 36:166–169Crossref, Google Scholar
36 : Long-term outcomes of office-based buprenorphine/naloxone maintenance therapy. Drug Alcohol Depend 2010; 106:56–60Crossref, Google Scholar
37 : Buprenorphine treatment for hospitalized, opioid-dependent patients: a randomized clinical trial. JAMA Intern Med 2014; 174:1369–1376Crossref, Google Scholar
38 : Computerized behavior therapy for opioid-dependent outpatients: a randomized controlled trial. Exp Clin Psychopharmacol 2008; 16:132–143Crossref, Google Scholar
39 : Adding an Internet-delivered treatment to an efficacious treatment package for opioid dependence. J Consult Clin Psychol 2014; 82:964–972Crossref, Google Scholar
40 : Methadone versus buprenorphine with contingency management or performance feedback for cocaine and opioid dependence. Am J Psychiatry 2005; 162:340–349Crossref, Google Scholar
41 : Comparison of buprenorphine treatment for opioid dependence in 3 settings. J Addict Med 2012; 6:68–76Crossref, Google Scholar
42 : A Community Reinforcement Plus Vouchers Approach: Treating Cocaine Addiction. Rockville, Md, National Institute on Drug Abuse, 1998Google Scholar
43 : Contingent reinforcement increases cocaine abstinence during outpatient treatment and 1 year of follow-up. J Consult Clin Psychol 2000; 68:64–72Crossref, Google Scholar
44 : The Prevention and Treatment of Missing Data in Clinical Trials. Washington, DC, National Academies Press, 2010Google Scholar
45 : Long-term outcomes from the National Drug Abuse Treatment Clinical Trials Network Prescription Opioid Addiction Treatment Study. Drug Alcohol Depend 2015; 150:112–119Crossref, Google Scholar
46 : A 33-year follow-up of narcotics addicts. Arch Gen Psychiatry 2001; 58:503–508Crossref, Google Scholar
47 : Who benefits from additional drug counseling among prescription opioid-dependent patients receiving buprenorphine-naloxone and standard medical management? Drug Alcohol Depend 2014; 140:118–122Crossref, Google Scholar
48 : Interviewer severity ratings and composite scores of the ASI: a further look. Drug Alcohol Depend 1994; 34:201–209Crossref, Google Scholar
49 : Buprenorphine/naloxone and methadone maintenance treatment outcomes for opioid analgesic, heroin, and combined users: findings from starting treatment with agonist replacement therapies (START). J Stud Alcohol Drugs 2013; 74:605–613Crossref, Google Scholar
50 : A comparison of buprenorphine taper outcomes between prescription opioid and heroin users. J Addict Med 2013; 7:33–38Crossref, Google Scholar
51 : Cognitive behavioral therapy improves treatment outcomes for prescription opioid users in primary care buprenorphine treatment. J Subst Abuse Treat 2016; 71:54–57Crossref, Google Scholar
52 : Buprenorphine retention in primary care. J Gen Intern Med 2005; 20:1038–1041Crossref, Google Scholar
53 : Influence of psychotherapy attendance on buprenorphine treatment outcome. J Subst Abuse Treat 2005; 28:247–254Crossref, Google Scholar
54 : Treating opioid addiction with buprenorphine-naloxone in community-based primary care settings. Ann Fam Med 2007; 5:146–150Crossref, Google Scholar
55 : Integrating buprenorphine maintenance therapy into federally qualified health centers: real-world substance abuse treatment outcomes. Drug Alcohol Depend 2013; 131:127–135Crossref, Google Scholar
56 : What should we be aiming for in the treatment of addiction? Addiction 2009; 104:685–686Crossref, Google Scholar
57 : A qualitative study of the adoption of buprenorphine for opioid addiction treatment. J Subst Abuse Treat 2014; 46:390–401Crossref, Google Scholar
58 : A stepped care strategy using buprenorphine and methadone versus conventional methadone maintenance in heroin dependence: a randomized controlled trial. Am J Psychiatry 2007; 164:797–803Crossref, Google Scholar
59 : Optimizing psychosocial support during office-based buprenorphine treatment in primary care: patients’ experiences and preferences. Subst Abus 2016; 37:70–75Crossref, Google Scholar
60 : A national study of a telephone support service for patients receiving office-based buprenorphine medication-assisted treatment: study feasibility and sample description. J Subst Abuse Treat 2010; 39:307–317Crossref, Google Scholar
61 : Integrating buprenorphine treatment into office-based practice: a qualitative study. J Gen Intern Med 2009; 24:218–225Crossref, Google Scholar
62 : Behavioral contingencies improve counseling attendance in an adaptive treatment model. J Subst Abuse Treat 2004; 27:223–232Crossref, Google Scholar
63 : Initial response as a predictor of 12-week buprenorphine-naloxone treatment response in a prescription opioid-dependent population. J Clin Psychiatry 2015; 76:189–194Crossref, Google Scholar
64 : Improving the quality of addiction treatment, in Interventions for Addiction. Edited by Miller PM, Comprehensive Addictive Behaviors and Disorders. San Diego, Academic Press, 2013, pp 579–588Crossref, Google Scholar
65 : A policy-oriented review of strategies for improving the outcomes of services for substance use disorder patients. Addiction 2011; 106:2058–2066Crossref, Google Scholar
66 : Buprenorphine therapy for opioid addiction in rural Washington: the experience of the early adopters. J Opioid Manag 2012; 8:29–38Crossref, Google Scholar
67 : Patient satisfaction with primary care office-based buprenorphine/naloxone treatment. J Gen Intern Med 2007; 22:242–245Crossref, Google Scholar
68 : Primary care-based buprenorphine taper vs maintenance therapy for prescription opioid dependence: a randomized clinical trial. JAMA Intern Med 2014; 174:1947–1954Crossref, Google Scholar
69 : Mortality among regular or dependent users of heroin and other opioids: a systematic review and meta-analysis of cohort studies. Addiction 2011; 106:32–51Crossref, Google Scholar
70 : Clinical trials in psychiatry: should protocol deviation censor patient data? Neuropsychopharmacology 1992; 6:39–48Google Scholar
71 : Predicting long-term stable recovery from heroin addiction: findings from a 33-year follow-up study. J Addict Dis 2007; 26:51–60Crossref, Google Scholar
72 : The anatomy of a follow-up. Br J Addict 1992; 87:1327–1333Crossref, Google Scholar
73 : Achieving a 96.6 percent follow-up rate in a longitudinal study of drug abusers. Drug Alcohol Depend 1996; 41:209–217Crossref, Google Scholar



