Updates in the Assessment and Management of Agitation
Abstract
Agitated patients can be dangerous to themselves and others. In fact, severe medical complications and death can occur with severe agitation. Because of this, agitation is considered a medical and psychiatric emergency. Regardless of the treatment setting, identifying the agitated patient early is a necessary skill. The authors review relevant literature regarding the identification and management of agitation and summarize current recommendations for adults, as well as children and adolescents.
Agitation can be conceptualized as a vital sign and a harbinger of disequilibrium in the patient’s emotional and behavioral regulation due to physical, psychosocial, developmental, psychiatric, and psychological contributors. Agitation occurs along a spectrum and can progress to aggression, with many antecedent factors influencing its presentation. Evaluation and management can be challenging because of the heterogeneity of factors, the populations and settings affected by agitation, and the variable utilization of standardized, evidence-based practice in the management of agitation.
Agitation in the acute care setting has risen in magnitude as a topic of research and clinical management optimization. The importance of this topic has been particularly highlighted by the rise of workplace violence (1) and by increased lengths of stay, or “boarding,” in the emergency department (ED) (2, 3). Establishing general, evidence-based principles and approaches to the evaluation and management of agitation, including early identification and prevention, utilization of a conceptual framework for establishing the potential etiologies influencing agitation, and provision of expeditious interventions based on a sound etiologically informed management plan, is critical to maintaining a safe and therapeutic care environment (4). The consequences of mismanagement of agitation are numerous, including poor outcomes for patients and families, injury to staff, moral distress for care teams, and ineffective utilization of health care resources (5).
In 2012, the American Association for Emergency Psychiatry’s (AAEP’s) Project BETA laid out best practice guidelines for the evaluation and management of agitation, highlighting the importance of universal implementation of nonpharmacological interventions, such as environmental modification and verbal de-escalation strategies, as well as providing a framework for principled, etiologically focused pharmacological strategies (6). In the subsequent decade, research and clinical experience have tested, challenged, and expanded those guidelines and have set forth new recommendations, including a similar pediatric consensus guideline developed by Gerson et al. in 2019 (7). Recent literature has focused on management of agitation in specific populations, novel agents for agitation, and reconsideration of previous medications (e.g., droperidol), in addition to exploring issues of equity and bias in the management of agitation. This article reviews current literature in order to summarize the most contemporary recommendations for agitation evaluation and management and also outlines the challenges that the heterogeneity in the presentations and causes of agitation can pose in the development of unifying recommendations.
Assessment
Two important factors in managing patients with agitation are early recognition and detection of risk, as well as pairing that understanding with specific interventions targeted to the etiologies driving the presentation of agitation (6, 8). Early awareness of risk factors and detection of risk with screening instruments can aid in early identification of the at-risk patient and prevent injury and restraint use (9, 10).
Risk Factors and Acute Presentations
Common risk factors for agitation (6, 9, 11, 12) and signs and symptoms of acute clinical presentations (9, 13, 14) are described in Box 1.
BOX 1. Modifiable and nonmodifiable risk factors for agitation
Nonmodifiable Risk Factors
History of agitation or violence
Previous psychiatric hospitalization
History of disciplinary action at school
Traumatic brain injury, developmental or cognitive delay
Borderline or antisocial personality disorder
History of sexual or physical abuse, interpersonal violence
Young, male, socially disadvantaged, psychosocial stressors
History of property destruction
Criminal history, gang involvement
Modifiable Factors
Acute clinical presentation
Physical illness
Substance use disorder
Psychiatric illness
Psychological factors (impulsivity, poor distress tolerance, limited insight, difficulty establishing trusting relationships, negative world view, lack of empathy, difficulty with authority figures)
Acute Clinical Presentations of Agitation
Inability to stay calm
Excessive purposeless motor or verbal activity, repetitive, restless
Paranoid, fearful, suspicious
Challenging, threatened by authority
Dysphoria, irritability, emotional lability, hostile mood
Escalating tone of voice, intense eye contact
Confused, disoriented, inattentive, impulsive, delirious
Intoxicated
Inability to communicate
Vital sign changes
Muscle tension, pacing
Screening and Rating Scales for Agitation
Current consensus does not value one screening tool over another (11). The choice of rating scale should be determined by best clinical fit for area, patient mix, time constraints, ease of use, and workflow. Commonly used screening tools (4, 9, 11, 15–18) are listed in Table 1. The AAEP’s Project BETA noted that the Behavioural Activity Rating Scale is easy to use even by individuals with limited or no training in its use (11). The utility of screening tools is often demonstrated in research, but use of such tools has limitations in busy high-acuity settings, such as the ED (19). These tools provide a snapshot of a patient’s current state, which may evolve over time, thus requiring frequent reassessment (9).
| Scale and setting | Type of screening | Comments |
|---|---|---|
| Psychiatric emergency service and emergency department (ED) | Quick screening | High-acuity setting |
| Agitated Behavior Scale (15, 16) | 14 items rated on a 4-point scale of present to absent | Clinician rated, developed for traumatic brain injury, can be used in the ED |
| Behavioural Activity Rating Scale (4, 11) | Single descriptor, seven levels (agitated to sedated) | Observational, easy to use in nonpsychiatric settings or ED |
| Sedation Assessment Tool (17) | 7-point scale of agitation to sedation in two descriptors (speech and responsiveness) | Quick, adaptation of altered mental status score, can be used in the ED |
| Inpatient setting | Longitudinal screening | Inpatient and long-term care |
| Aggressive Behavior Scale (15) | Four-item summary scale | Scored over 7 days, best for inpatient and long-term care |
| Cohen-Mansfield Agitation Inventory (9) | 29 behaviors rated on a 7-point severity scale | Labor intensive, scored over 2 weeks, caregiver rated |
| Richmond Agitation Sedation Scale (4, 18) | 10-point scale (agitation to sedation) in two descriptors | Quick delirium screen for critical care setting, repeat over time |
TABLE 1. Summary of screening tools for agitation in adults
Etiologies Influencing Agitation
The determination of etiology or etiologies contributing to the presentation of agitation will best inform targeted agitation management strategies. It is critical for the astute clinician not simply to view agitation in a singular lens and utilize rote management strategies based on provider comfort; rather, one should customize management to the unique patient’s needs based on a thoughtful biopsychosocial assessment of factors precipitating and perpetuating the presentation of agitation. Because of limitations on time, information, and patient engagement, expeditious assessment and identification of an underlying medical problem are necessary (9, 19).
At the initial presentation and triage, basic vital signs and pertinent lab tests should be obtained. Direct observation of the patient’s presentation and brief assessment of mental status often are the highest yield (4). Lab testing should be targeted, focusing on suspected etiology and common antagonizing factors, rather than universal screening labs (11). Point-of-care glucose and oxygen saturation should be conducted, as well as a urine drug screen on notably impaired or agitated patients, particularly when intoxication is suspected but not confirmed by history (4, 9, 11). Patients with suspected delirium should receive a focused examination with vital signs to localize potential sources of systemic illness to then guide diagnostic assessment, which may include a comprehensive metabolic panel, complete blood count, urinalysis, urine drug screen, and often head imaging. Additional collateral information to aid in assessment should be gathered quickly and efficiently. This can include verbal reports from law enforcement, emergency medical services, family, partners, referral sources, outpatient providers, and current care team members. Agitation is a medical emergency, and information needs to be gathered regardless of confidentiality concerns and patient consent (19). Medical records should be reviewed, even if only a cursory review can be conducted, to determine psychiatric and medical history, establish a baseline, and assess for recent substance use and intoxication (4, 9, 19).
Physical illness and delirium.
Several indicators may suggest a medical etiology. According to Nordstrom et al. (11), “new onset of agitation in a person older than 45 with no psychiatric history is suggestive of a medical etiology.” Signs and symptoms of medical etiologies of agitation in the acute presentation include abnormal vital signs, abnormal labs, no past psychiatric history, fluctuation in attention and awareness, changes in cognition or orientation, focal physical examination findings, and clear localization of pain (11, 20). Agitation of unknown etiology should be considered to have a medical cause until proven otherwise (9, 11). This should be particularly considered in the nonverbal patient, where expressions of discomfort or pain may be manifested as agitation. Medical conditions associated with agitation are outlined in Table 2 (4, 9, 11, 20, 21). Delirium, or impairment in attention and awareness with a fluctuating course and associated cognitive disturbances, is frequently associated with agitation, results from physical disease, and can often be missed on initial assessment, with potential catastrophic consequences (8, 11, 19).
| Type | Examples |
|---|---|
| Neurological | Traumatic brain injury, intracranial hemorrhage, seizure, cardiovascular accident, transient ischemic attack, encephalopathies (such as hepatic or renal), autoimmune encephalitis |
| Infectious | Encephalitis, meningitis, sepsis, urinary tract infection or urosepsis (elderly), pneumonia |
| Gastrointestinal or metabolic | Electrolyte disturbances, hypoglycemia, diabetic ketoacidosis, ileus, inflammatory bowel disease, chronic malnutrition, gastrointestinal obstruction |
| Respiratory | Hypoxia, pulmonary embolism, respiratory failure |
| Cardiac | Arrhythmia, myocardial infarction, heart failure |
| Toxicology | Environmental toxins, medication reactions, neuroleptic malignant syndrome, serotonin syndrome |
| Endocrine | Thyroid (thyrotoxicosis, myxedema) |
| Hematologic or oncologic | Anemia, oncologic process |
| Constitutional | Poor sleep, pain, hypothermia, or hyperthermia |
TABLE 2. Common medical etiologies of agitation
Substance use and withdrawal.
Substance intoxication and withdrawal are frequent causes of agitation, especially in the emergency and hospital setting, and substance use is an important consideration when selecting medication to treat agitation. For many substances, both intoxication and withdrawal states can contribute to agitation (9, 11, 20–22). Withdrawal from alcohol or benzodiazepines can be life threatening, making early awareness vital to management (21). Methamphetamine-related presentations linked to increased agitation and aggression, combined with increased and widespread use, accounted for more than 2% of all ED presentations globally (23, 24).
Psychiatric and developmental conditions.
Several psychiatric diagnoses are known to have an increased risk of agitation (9, 11, 20, 25, 26), especially at times of acute exacerbation. Awareness of prior psychiatric history, onset of symptoms, and baseline level of function are important factors in appropriate management (25).
Management
Management of a patient presenting with agitation consists of three components: nonpharmacological interventions (such as verbal de-escalation, change in environmental factors, and psychoeducation), medications, and physical restraint. The goal of intervention is to calm the patient in the least invasive way, without causing oversedation. If medication is indicated, the de-escalation process should include coupling nonpharmacological techniques with the offer of targeted medication on the basis of both patient preference and suspected etiology. Once calmed, the patient should be iteratively assessed to provide ongoing information to enhance longitudinal management. Figure 1 outlines decision points for each intervention.
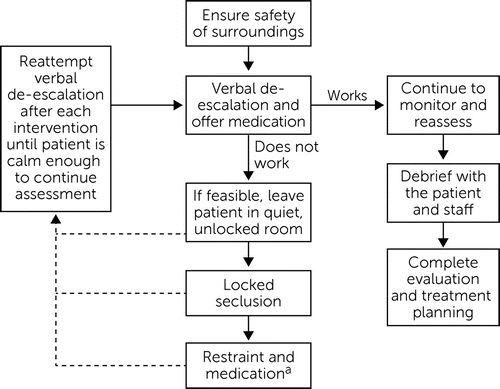
FIGURE 1. Decision points for management of a patient presenting with agitation
aOral medication is favored over intramuscular medication.
Nonpharmacological Interventions
Disagreement with staff, pain and discomfort, and perceived failure to address needs are common precipitants of agitation (27). Environmental factors can also play a role in agitation. Crowding, bright lights, increased wait times, and noise pollution provoke agitation (20). The physical space should feel safe for the patient and staff. When possible, moving the patient to a low-stimulation environment can aid in de-escalation (14, 28).
Verbal de-escalation remains the first-line treatment. Staff should be trained in de-escalation techniques and be aware of their own countertransference to avoid provoking the patient further. Ideally, four to six team members should be present to engage the patient, both to discourage violent behavior and to have enough staff present if de-escalation attempts fail and restraint is necessary (14). This should be balanced with an awareness of how increased staff presence may escalate a patient’s agitation, particularly for individuals with significant trauma histories, autism, or developmental delay.
Communication skills for verbal de-escalation, on the basis of expert consensus, are described in Table 3 (14, 28, 29). Patients should be engaged with an open posture, with hands visible and relaxed. Eye contact should be casual and nonthreatening (14). Validation, reassurance, appropriate and skillful use of humor, and reframing may help to defuse a tense situation. If verbal de-escalation is successful, continual reassessment is recommended to monitor for signs of re-emerging agitation (28). After an episode of agitation, especially episodes involving involuntary medication or restraint, debrief with the patient and staff to reflect on understanding the factors influencing agitation and response to treatment (14).
| Skill | Comments |
|---|---|
| Respect personal space | Maintain at least two arms’ lengths (6 feet) and allow space for both you and patient to exit |
| One person | Designate one person to engage the patient; introduce yourself and your role |
| One message | Be concise, speak clearly and simply, repeat instructions multiple times |
| Name feelings | Listen closely to try to identify and validate the patient’s emotional experience; share how the patient’s behavior is affecting you; reflect what you are hearing to clarify emotions and meaning behind statements shared |
| Appeal to the patient’s wants or needs | Listen for anything you may be able to offer the patient, such as a psychotherapeutic intervention or a comfort item; focus on what you can do for the patient |
| Set limits and reinforce appropriate behavior | Respectfully communicate your expectations and the consequences of unacceptable behaviors; focus on shared values, such as safety, respect, and improvement in the patient’s quality of life or functioning |
| Offer choices | Ask the patient to consider what has worked in the past; offer choices between medications or route of administration |
TABLE 3. Communication skills for verbal de-escalation
Pharmacological Management
Sometimes verbal de-escalation is sufficient to calm the patient, especially when the source issue is addressed (hunger, overstimulation, pain). When the source still exists, coupling de-escalation with medication is key. Whenever it is safe to do so, offer oral medication first and involve the patient in shared decision making regarding choice of medication. Patients often have a preference or seek to avoid medications, on the basis of previous experience, and their perspective can be invaluable to selecting an appropriate regimen and avoiding more coercive measures.
Oral and intramuscular (IM) medications have similar efficacy, with IM formulations having quicker onset of action (8, 30). Table 4 describes medications used in the treatment of agitation (8, 9, 28, 30–34). When selecting a medication, consider the patient’s physical health status (including nutrition and renal and hepatic function), preferences, known comorbid conditions, and any past reactions to medications. In terms of the medication itself, be mindful of the route of delivery, onset and duration of action, and anticipated side effects. Patient- and medication-related factors should then be paired with a working conceptualization of the etiologies potentially affecting the patient’s presentation.
| Suspected etiology | Medication and dose | Comments |
|---|---|---|
| Primary psychiatric or undifferentiated with prominent psychosis | Olanzapine, 5 mg–10 mg (PO, ODT or IM); risperidone, 2 mg (PO, ODT or liquid); ziprasidone, 10 mg–20 mg (IM); haloperidol, 2 mg–10 mg (PO or IM); droperidol, 5 mg–10 mg (IM) | Add lorazepam 1 mg–2 mg if haloperidol or monotherapy ineffective; risk of respiratory depression for IM olanzapine and IM lorazepam within 1 hour |
| Intoxication with central nervous system depressant (including alcohol) | Haloperidol, 2 mg–10 mg (PO or IM); olanzapine, 5 mg (PO); risperidone, 2 mg (PO, ODT or liquid) | Avoid benzodiazepines if possible; FGAs still heavily favored, but SGAs likely safe if given PO; IM risks respiratory depression |
| Stimulant intoxication, alcohol or benzodiazepine withdrawal OR undifferentiated without psychosis | Lorazepam, 1 mg–2 mg (PO or IM); diazepam, 5 mg–10 mg (PO); midazolam, 5 mg–10 mg (IM) | Add SGA if prominent psychotic symptoms |
| Delirium | Risperidone, 1 mg–2 mg (PO, ODT); olanzapine, 5 mg–10 mg (PO, ODT or IM); haloperidol, <5 mg (PO or IM); ziprasidone, 10 mg–20 mg (IM) | Avoid benzodiazepines; high risk of EPS with haloperidol doses >3 mg; consider QTc monitoring |
TABLE 4. Summary of medications for management of agitation in adults, by suspected etiologya
Antipsychotics.
First-line pharmacological agents are benzodiazepines and antipsychotics, depending on the underlying etiology of agitation (8, 9, 28). In general, antipsychotic medication is preferred in cases of delirium, agitation due to alcohol intoxication, agitation due to a known psychiatric disorder, and undifferentiated presentations with prominent psychotic symptoms (8). Antipsychotics are relatively contraindicated in the management of catatonia.
Second-generation antipsychotics (SGAs), such as olanzapine, ziprasidone, and risperidone, are favored over first-generation antipsychotics (FGAs), such as haloperidol and droperidol. Both antipsychotic classes have similar efficacy in treating agitation, but FGAs have more adverse effects, namely extrapyramidal symptoms (EPS) and QTc prolongation, with the associated increased risk of arrhythmia (8, 35). Use caution in geriatric patients and patients in early childhood with delirium as these populations are particularly susceptible to QTc prolongation and EPS. To minimize EPS, haloperidol is often administered in combination with a benzodiazepine or diphenhydramine or both. Data supporting the need for this practice are limited, and the combination of three agents (haloperidol, lorazepam, and diphenhydramine) risks oversedation. Even with EPS-mitigating medications, the emergence of EPS can be delayed for longer than 24 hours (32). Droperidol, once avoided due to its black box warning for QTc prolongation, has seen a resurgence of use for agitation in emergency settings, particularly because of its quick onset of action. Doses of up to 10 mg have been found to be as safe as other medications used in the treatment of agitation (36). When used for agitation in patients with alcohol intoxication, it is associated with a shorter length of stay, compared with olanzapine or haloperidol (37).
Of the SGAs, olanzapine has the strongest evidence base for treatment of agitation, with good efficacy and tolerability for oral and IM formulations. IM olanzapine has higher alpha-2 effect in this formulation and should be avoided within 1 hour of IM or intravenous (IV) benzodiazepines because of the risk of respiratory depression (8). Alternatives to olanzapine with good supportive data include oral risperidone and IM ziprasidone (8, 9). SGAs still have some risk of QTc prolongation, but the risk is lower than for FGAs, and cases of torsades de pointes are rare (8, 35).
Both FGAs and SGAs have a black box warning for their association with increased risk of mortality in elderly patients with dementia (38). As such, the risks and benefits should be weighed carefully, and the patient and those involved in the patient’s care should be educated on the risks (39).
Benzodiazepines.
Benzodiazepines are favored in cases of agitation due to alcohol or benzodiazepine withdrawal, intoxication with a stimulant, or in undifferentiated agitation without prominent psychosis (8). In a study comparing IM midazolam to antipsychotics, midazolam was faster acting than haloperidol or ziprasidone, but it often required repeat dosage (34). Expert consensus favors oral and IM lorazepam (8), which has a slower onset but longer duration of action. Benzodiazepines may worsen delirium or cause respiratory depression in patients with respiratory conditions or those intoxicated with alcohol or central nervous system depressants.
Other medications or formulations.
Research into novel agents for the treatment of agitation continues to evolve. Inhaled loxapine (28, 40) and dexmedetomidine film (41) are approved by the Food and Drug Administration for treatment of agitation in bipolar disorder and schizophrenia. Ketamine is used widely in IM and IV formulations in prehospital and emergency settings, but intranasal ketamine has not been trialed in this population (28, 42). Although some practitioners prefer these agents, there is not sufficient evidence to recommend them over the medications listed in Table 4.
Inhaled loxapine, an FGA, is a novel route of administration, with onset of action similar to that of IV medication (10 minutes). The inhaled formulation has lower risk of systemic side effects, such as EPS, but carries a risk of bronchospasm in patients with respiratory conditions. Ketamine is an NMDA-receptor antagonist that causes dissociation and sedation. It has rare complications of hypersalivation, vomiting, and laryngospasm; it also lacks antipsychotic properties and has a higher risk of oversedation, compared with antipsychotics. Ketamine can cause psychotic symptoms when the patient emerges from sedation, which has prompted some to jointly administer ketamine with a benzodiazepine (28). Dexmedetomidine is a selective alpha-2 receptor agonist that has sedative but not antipsychotic properties. The sublingual film has had positive results in treating mild to moderate agitation, with rare side effects of somnolence, dry mouth, dizziness, and hypotension and no serious adverse effects observed in trials (41).
Restraint and Seclusion
Restraint or seclusion is considered a last resort when less restrictive options are ineffective to ensure safety. If a patient can maintain safety in a locked room and one is available, seclusion is favored over physical restraint (43). Because patients are restrained or secluded involuntarily, state and organizational guidelines for involuntary treatment must be followed. The Joint Commission (44) advises that restraint or seclusion episodes must be:
Performed by trained staff,
Implemented only after less restrictive options have failed,
Initiated for safety and not as a means to punish or retaliate,
Monitored continuously with a goal to discontinue as soon as possible, and
Clearly documented (including reason, monitoring practices, patient response, and indications to discontinue).
Use of restraints is associated with significant morbidity and mortality. Death has occurred due to strangulation from improper placement or due to other complications, such as rhabdomyolysis, thrombosis, positional asphyxia, or blunt force trauma (45). Restraint episodes are distressing and dehumanizing and can result in long-lasting trauma and distrust of the health care system (45–47). Incidence of posttraumatic stress disorder following restraint and seclusion ranges from 25% to 47% (47). Restraint episodes put staff at risk of physical and psychological harm (43) and are subject to bias. Recent studies found that Black, male, and homeless patients are more likely to be restrained (48, 49). Jin and colleagues (50) have proposed recommendations to mitigate bias in caring for agitated patients. At institutional and systems levels, they propose increasing support for focused research, improving staffing and overcrowding in EDs, and improving bias and restraint education. On an individual level, they recommend the following five questions to assess whether bias is affecting one’s care of an agitated patient:
Have I tried verbal de-escalation techniques, listened to the patient’s desires, and offered alternatives to restraint?
Would another staff member be better at de-escalating this patient based on demographic similarities or perceived patient preference (i.e., a patient who responds better to female staff)?
Is my fear of this patient influenced by the patient’s appearance?
Is the patient’s perception of control or way of expressing frustration culturally different from my own?
Am I using racial, gender, socioeconomic, or another harmful bias to guide my care of this patient?
Evaluation and Management of Agitation in Youths
Adolescents may present to the ED with agitation or become agitated in the ED. One report noted that one in 15 youths who presented to the ED with a psychiatric chief complaint were restrained, often due to agitation, and that youths with autism spectrum disorder (ASD) had a higher prevalence of restraint (51–53). Risk factors for agitation among youths are incorporated in Box 1 and Table 2.
Evaluation
Determining the etiology of agitation is of paramount importance. Evaluation of agitation in at-risk youths begins with heightened awareness of early signs of its preceding presentation, its evolution, and the associated risk factors. Evaluation should incorporate understanding of development, family-related factors, and systems of care that may affect the care experience of youths (54). Specific screening tools utilized among youths (55–59) are identified in Table 5.
| Screening tool | Description | Comments |
|---|---|---|
| Abbreviated Brief Rating of Aggression by Children and Adolescents (55) | 14-item instrument | Used to predict potential for agitation and aggression on an inpatient unit |
| Dynamic Appraisal of Situational Aggression (inpatient) (56, 57) | Seven items rated present or absent | Assesses likelihood of aggression in the next 24 hours |
| Modified Overt Aggression Scale (58, 59) | Four-part behavioral rating scale; four types of agitation or aggression rated over past week | Inpatient or residential setting |
TABLE 5. A summary of screening tools for agitation in youths
Observation of the interactions between the caregiver and the child provide information about potential causes of agitation. It is important to keenly monitor how the caregiver responds to the child’s distress, how the child displays and communicates that distress, the sense of secure attachment between the child and the caregiver, and the reinforcing or mitigating factors affecting the delicate interplay between the child and caregiver (7). Gathering history involves interviewing the child and the child’s caregivers, initially together and then separately (60). The age and developmental status of the child drives the decision of the sequencing of the interviews and may inform whether the child is able to separate meaningfully from the caregiver (61). When selecting whom to interview first alone, consider interviewing adolescent patients alone to honor their autonomy and highlight the value of their report, which can aid in rapport building (62). Further collateral may need to be gleaned from specific sources unique to youths.
The interviewer should be mindful to obtain thoughtful birth, developmental, social, family, and early childhood histories. Interviewing the child requires a strong understanding of the child’s development, cognitive status, and evolving language use and levels of insight. Key clinical pearls for interviewing youths (54) are described below:
Temporal relationships are less reliable in younger children; association of events may need to be tied to life events (e.g., holidays, school occasions, and birthdays).
Concrete, simplistic conversations regarding sexual behaviors, substance use, and suicide may be needed in preteen populations.
Caregivers’ and youths’ reports may differ; youths tend to be more accurate in reporting internalizing symptoms, and caregivers are more valid in their observations of externalizing symptoms, such as agitation.
Interviews can start with the child and family together but then should transition to a private interview with the adolescent alone first, to emphasize the developmentally appropriate desire for individuation, or may proceed with the parent alone first when the child is a preteen.
Look for signs of trauma and self-injury in youths exhibiting new-onset agitation or agitation without clear cause.
Assess for dysmorphism or congenital abnormalities, while conducting a brief neurologic assessment to identify any factors that may suggest a genetic or neurodevelopmental disorder.
Youths have limited capacity to verbalize distress, which warrants increased awareness and surveillance for potential sources of pain (e.g., dental disease, constipation, fractures, and skin injury), as well as other sources of discomfort (e.g., poor sleep, hunger, and metabolic or endocrine disorders).
Youths with eating disorders, dehydration, or poor nutrition may have electrolyte abnormalities that are important to assess and monitor.
Nonpharmacological Interventions
Management of agitation in youths involves even greater focus on prevention, early nonpharmacological intervention, and environmental modification, which may mitigate the use of psychopharmacology (7). The use of standardized protocols and developmentally appropriate, trauma-informed, and family-centered care practices aid in de-escalation. Involvement of support staff trained in de-escalation, distraction, and comfort measures, such as Child Life, should also be considered (7, 54, 63).
As noted previously, a thoughtful environment is a powerful way to promote a therapeutic atmosphere. Access to certain sensory stimulation that is soothing, such as a weighted blanket, may be highly effective in promoting calm or de-escalating an irritable child (63). Recognizable objects from home, as well as the availability of support family, can be very comforting to the child exhibiting agitation. Use of a token economy, such a sticker chart, can also be a helpful tool in reinforcing positive behaviors (64). It is important to address developmental needs that can exacerbate risk of aggression due to added frustration, barriers to communication, or reduction of the child’s sense of control in a situation. These factors include communication challenges and sensory limitations, as well as delays in meeting activities of daily living (63).
Pharmacological Management
Principles of pharmacological management of agitation in youths are similar to those described previously for adults, with a few key considerations specific to youths. A more extensive review of consensus guidance on the pharmacological management of agitation in youths is provided in the Pediatric BETA Guidelines by Gerson et al. in 2019 (7). Key recommendations from that study are summarized in Table 6 and Table 7. Special consideration is given to management of youths with autism and intellectual disability. Of note, Table 6 and Table 7 highlight the use of alpha-2 agonists and antihistamines. Although clonidine (an alpha-2 agonist) and diphenhydramine (an antihistamine) are highlighted here, guanfacine and hydroxyzine may also be utilized with good effect and may be considered if there is lack of response or oversedation with the use of clonidine or diphenhydramine (65–69).
| Medication | Dose | Peak effect (minutes) | Maximum daily dose (mg by weight or age) | Other considerations |
|---|---|---|---|---|
| Clonidine | PO, 0.05 mg–0.1 mg | 30–60 | <40 kg, 0.2 mg; >40 kg, 0.3 mg– 0.4 mg | Monitor for hypotension and bradycardia |
| Diphenhydramine | PO or IM, 12.5 mg– 50 mg or 1 mg per kg body weight | 120 | Age <12, 50 mg– 100 mg; age >12, 100 mg–200 mg | Avoid in delirium and in youths at risk of paradoxical reaction (e.g., autism); can use to mitigate EPS risk from antipsychotics |
| Lorazepam | PO, IM, or IV, 0.5 mg– 2 mg or 0.05 mg– 0.1 mg per kg body weight | PO, 60–120; IM or IV, 10 | Age <12, 4 mg; age >12, 6 mg– 8 mg | Avoid in delirium and in youths at risk of paradoxical reaction (e.g., autism); can use to mitigate EPS risk from antipsychotics |
| Chlorpromazine | PO or IM, 12.5 mg– 60 mg or 0.55 mg per kg body weight | PO, 30; IM, 15 | Age <5, 40 mg; age >5, 75 mg | Monitor for hypotension; IM dose half of PO dose |
| Haloperidol | PO or IM, 0.5 mg– 5 mg or 0.55 mg per kg body weight | PO, 30; IM, 15 | <40 kg, 6 mg; >40 kg, 15 mg | Monitor for hypotension; monitor EKG, particularly with IM or IV use; IM dose half of PO dose; EPS risk lower with IV use |
| Olanzapine | PO or IM, 2.5 mg– 10 mg | PO, 240–480; IM, 15–45 | Age > 12, 20 mg | ODT formulation available; do not give IM within 1 hour of IM or IV benzodiazepines |
| Risperidone | PO, 0.25 mg–1 mg or 0.005 mg–0.01 mg per kg body weight | 30–60 | Age <12, 1 mg–2 mg; age >12, 2 mg–4 mg | ODT formulation available; watch for EPS |
| Quetiapine | PO, 25 mg–50 mg or 1 mg–1.5 mg per kg body weight | 30 | Age >12, 600 mg– 800 mg | Monitor for sedation and hypotension |
TABLE 6. Medications commonly used in the management of agitation in youthsa
| Etiology | Evaluation considerations | Pharmacological management |
|---|---|---|
| Delirium | Regular medical evaluation; environmental modification | Avoid benzodiazepines and anticholinergics; PO, clonidine, quetiapine, risperidone, and olanzapine; IM or IV, olanzapine and haloperidol |
| Substance intoxication or withdrawal | History, physical examination, urine toxicology to guide understanding of potential substances implicated | Alcohol or benzodiazepine withdrawal, lorazepam with or without haloperidol; alcohol or benzodiazepine intoxication, haloperidol or chlorpromazine; opiate withdrawal, clonidine, opiate replacement, or supportive treatment; stimulant or phencyclidine intoxication, lorazepam with or without haloperidol |
| Developmental delay or autism | Behavioral intervention; assess for sources of pain or discomfort; support communication; address sensory factors | Benzodiazepines, antihistamines may cause paradoxical response; PO, alpha-2 agonist or antipsychotic; IM, olanzapine or chlorpromazine |
| Primary psychiatric diagnosis | Clarify diagnostic formulation | Catatonia, lorazepam; anxiety or trauma, lorazepam or clonidine; ADHD, alpha-2 agonist, antihistamine, or antipsychotic; oppositional defiant or conduct disorder, antipsychotic or lorazepam; mania or psychosis, antipsychotic with lorazepam or diphenhydramine if concern for EPS |
| Unknown or mixed etiology | Reinforce environmental and behavioral strategies while searching for etiologic factors | Mild or moderate, diphenhydramine, lorazepam, or olanzapine; moderate or severe, chlorpromazine, haloperidol, olanzapine, or lorazepam |
TABLE 7. Summary of medications for management of agitation in youths, by etiologya
Pharmacological management begins with a careful review of current and past prescribed home medications, including over-the-counter, alternative, and dietary products, given the higher propensity of medication sensitivities or side effects in some youths (70, 71). In particular, antihistamines and benzodiazepines can cause excitation or paradoxical worsening of agitation, and whether this occurs should be asked routinely in patients, particularly younger children, youths with developmental delay, and youths with autism (7, 54). Furthermore, current medication use should include review for drug-drug interactions that may result in avoidable toxicities in youths (72). Often a half or extra dose of an effective, previously prescribed, psychotropic medication is the best approach (7, 73, 74). Lower doses and slower titration may be required to reach therapeutic effect while limiting side effects, when the acuity of presentation does not warrant more rapid titration (75, 76). In general, preteen patients less than 40 kg in weight will require weight-based dosing, and adolescents, often greater than 40 kg in weight, will receive doses similar or slightly reduced, compared with adult doses (77). Given the greater variability in response, tolerability and efficacy should be routinely assessed, and dosing should be adjusted accordingly. Generic agents are safe and effective, yet the pharmacokinetic bioequivalence of generic compounds ranges from 80% to 125%, compared with brand-name drugs (78). Thus, switching between brand and generic medication can change the efficacy or produce side effects, particularly in medically ill youths, younger youths, or youths with developmental disorders who may be more sensitive to these changes (79).
Future Directions
Since the development of the Project BETA guidelines, more focus has been placed on the management of agitation, especially on novel agents such as inhaled loxapine, ketamine, and dexmedetomidine. It is equally important for the field to better understand the role of nonpharmacological measures. For some, the idea of de-escalating by offering choices, making environmental changes, or simply talking with the patient (using basic techniques) appears too simple. Until studies support these measures, they may continue to appear to be the “art” of psychiatry, rather than teachable practices. We also need to better understand the role of racial and ethnic biases in our care of patients to ensure equitable and humane treatment, no matter the skin color, language, religion, or other differences.
1 ACEP Emergency Department Violence Poll Research Results. Alexandria, VA, Marketing General Incorporated, 2018. https://www.emergencyphysicians.org/globalassets/files/pdfs/2018acep-emergency-department-violence-pollresults-2.pdfGoogle Scholar
2 : Boarding of mentally ill patients in emergency departments: American Psychiatric Association resource document. West J Emerg Med 2019; 20:690–695Crossref, Google Scholar
3 : Worsening of emergency department length of stay during the COVID-19 pandemic. J Am Coll Emerg Physicians Open 2021; 2:e12489Google Scholar
4 : Approach to the agitated emergency department patient. J Emerg Med 2018; 54:447–457Crossref, Google Scholar
5 : Workplace violence against health care workers in the United States. N Engl J Med 2016; 374:1661–1669Crossref, Google Scholar
6 : Overview of Project BETA: Best Practices in Evaluation and Treatment of Agitation. West J Emerg Med 2012; 13:1–2Crossref, Google Scholar
7 : Best Practices for Evaluation and Treatment of Agitated Children and Adolescents (BETA) in the emergency department: consensus statement of the American Association for Emergency Psychiatry. West J Emerg Med 2019; 20:409–418Crossref, Google Scholar
8 : The psychopharmacology of agitation: consensus statement of the American Association for Emergency Psychiatry Project BETA Psychopharmacology Workgroup. West J Emerg Med 2012; 13:26–34Crossref, Google Scholar
9 : Assessment and management of agitation in psychiatry: expert consensus. World J Biol Psychiatry 2016; 17:86–128Crossref, Google Scholar
10 : A quality improvement project on agitation management in the emergency department. J Emerg Nurs 2021; 47:390–399.e3Crossref, Google Scholar
11 : Medical evaluation and triage of the agitated patient: consensus statement of the American Association for Emergency Psychiatry Project BETA Medical Evaluation Workgroup. West J Emerg Med 2012; 13:3–10Crossref, Google Scholar
12 : Managing agitation associated with schizophrenia and bipolar disorder in the emergency setting. West J Emerg Med 2016; 17:165–172Crossref, Google Scholar
13 : 1st International Experts’ Meeting on Agitation: conclusions regarding the current and ideal management paradigm of agitation. Front Psychiatry 2018; 9:54Crossref, Google Scholar
14 : Verbal de-escalation of the agitated patient: consensus statement of the American Association for Emergency Psychiatry Project BETA De-escalation Workgroup. West J Emerg Med 2012; 13:17–25Crossref, Google Scholar
15 : Systematic reviews of assessment measures and pharmacologic treatments for agitation. Clin Ther 2010; 32:403–425Crossref, Google Scholar
16 : Psychometric testing of the Agitation Severity Scale for acute presentation behavioral management patients in the emergency department. Adv Emerg Nurs J 2014; 36:250–270Crossref, Google Scholar
17 : Sedation Assessment Tool to score acute behavioural disturbance in the emergency department. Emerg Med Australas 2011; 23:732–740Crossref, Google Scholar
18 : Detecting delirium superimposed on dementia: evaluation of the diagnostic performance of the Richmond Agitation and Sedation Scale. J Am Med Dir Assoc 2016; 17:828–833Crossref, Google Scholar
19 : Psychiatric evaluation of the agitated patient: consensus statement of the American Association for Emergency Psychiatry Project Beta Psychiatric Evaluation Workgroup. West J Emerg Med 2012; 13:11–16
20 : Stabilization and management of the acutely agitated or psychotic patient. Emerg Med Clin North Am 2015; 33:739–752Crossref, Google Scholar
21 : A modern-day fight club? The stabilization and management of acutely agitated patients in the emergency department. Psychiatr Clin North Am 2017; 40:397–410Crossref, Google Scholar
22 : Psychiatric comorbidity associated with synthetic cannabinoid use compared to cannabis. J Psychopharmacol 2016; 30:1321–1330Crossref, Google Scholar
23 : Rates and features of methamphetamine-related presentations to emergency departments: an integrative literature review. J Clin Nurs 2018; 27:2569–2582Crossref, Google Scholar
24 : Prevalence and risk factors of emergency department presentations with methamphetamine intoxication or dependence: a systematic review and meta-analysis. Issues Ment Health Nurs 2019; 40:567–578Crossref, Google Scholar
25 : The agitated older adult in the emergency department: a narrative review of common causes and management strategies. J Am Coll Emerg Physicians Open 2020; 1:812–823Crossref, Google Scholar
26 : Characterizing the experience of agitation in patients with bipolar disorder and schizophrenia. BMC Psychiatry 2018; 18:104Crossref, Google Scholar
27 : Characteristics and severity of agitation associated with use of sedatives and restraints in the emergency department. J Emerg Med 2019; 57:611–619Crossref, Google Scholar
28 : Joint BAP NAPICU evidence-based consensus guidelines for the clinical management of acute disturbance: de-escalation and rapid tranquillisation. J Psychopharmacol 2018; 32:601–640Crossref, Google Scholar
29 : Preventing and de-escalating aggressive behavior among adult psychiatric patients: a systematic review of the evidence. Psychiatr Serv 2017; 68:819–831Crossref, Google Scholar
30 : Improving the management of acutely agitated patients in the emergency department through implementation of Project BETA (Best Practices in the Evaluation and Treatment of Agitation). J Am Coll Emerg Physicians Open 2020; 1:898–907Crossref, Google Scholar
31 : Randomized double‐blind trial of intramuscular droperidol, ziprasidone, and lorazepam for acute undifferentiated agitation in the emergency department. Acad Emerg Med 2021; 28:421–434Crossref, Google Scholar
32 : Efficacy of combination haloperidol, lorazepam, and diphenhydramine vs combination haloperidol and lorazepam in the treatment of acute agitation: a multicenter retrospective cohort study. J Emerg Med 2022; 62:516–523Crossref, Google Scholar
33 : A prospective study of intramuscular droperidol or olanzapine for acute agitation in the emergency department: a natural experiment owing to drug shortages. Ann Emerg Med 2021; 78:274–286Crossref, Google Scholar
34 : Intramuscular midazolam, olanzapine, ziprasidone, or haloperidol for treating acute agitation in the emergency department. Ann Emerg Med 2018; 72:374–385Crossref, Google Scholar
35 : QTc prolongation, torsades de pointes, and psychotropic medications. Psychosomatics 2013; 54:1–13Crossref, Google Scholar
36 : American Academy of Emergency Medicine position statement: safety of droperidol use in the emergency department. J Emerg Med 2015; 49:91–97Crossref, Google Scholar
37 : Parenteral antipsychotic choice and its association with emergency department length of stay for acute agitation secondary to alcohol intoxication. Acad Emerg Med 2019; 26:79–84Google Scholar
38 : Risk of mortality among individual antipsychotics in patients with dementia. Am J Psychiatry 2012; 169:71–79Crossref, Google Scholar
39 : The American Psychiatric Association practice guideline on the use of antipsychotics to treat agitation or psychosis in patients with dementia. Am J Psychiatry 2016; 173:543–546Crossref, Google Scholar
40 : Inhaled loxapine for the management of acute agitation in bipolar disorder and schizophrenia: expert review and commentary in an era of change. Drugs R D 2019; 19:15–25Crossref, Google Scholar
41 : Effect of sublingual dexmedetomidine vs placebo on acute agitation associated with bipolar disorder: a randomized clinical trial. JAMA 2022; 327:727–736Crossref, Google Scholar
42 : Ketamine as a first-line treatment for severely agitated emergency department patients. Am J Emerg Med 2017; 35:1000–1004Crossref, Google Scholar
43 : Use and avoidance of seclusion and restraint: consensus statement of the American Association for Emergency Psychiatry Project BETA Seclusion and Restraint Workgroup. West J Emerg Med 2012; 13:35–40Crossref, Google Scholar
44 . Milwaukee, Crisis Prevention Institute, 2010. https://www.crisisprevention.com/CPI/media/Media/Resources/alignments/Joint-Commission-Restraint-Seclusion-Alignment-2011.pdfGoogle Scholar
45 : Morbidity and mortality associated with the utilization of restraints: a review of literature. Psychiatr Q 2013; 84:499–512Crossref, Google Scholar
46 : Experiences of individuals who were physically restrained in the emergency department. JAMA Netw Open 2020; 3:e1919381Crossref, Google Scholar
47 : Effects of seclusion and restraint in adult psychiatry: a systematic review. Front Psychiatry 2019; 10:491Crossref, Google Scholar
48 : Disparities in care: the role of race on the utilization of physical restraints in the emergency setting. Acad Emerg Med 2020; 27:943–950Crossref, Google Scholar
49 : Association of race/ethnicity and other demographic characteristics with use of physical restraints in the emergency department. JAMA Netw Open 2021; 4:e2035241Crossref, Google Scholar
50 : Exploring bias in restraint use: four strategies to mitigate bias in care of the agitated patient in the emergency department. Acad Emerg Med 2021; 28:1061–1066Crossref, Google Scholar
51 : The use of restraint for pediatric psychiatric patients in emergency departments. Pediatr Emerg Care 2004; 20:151–156Crossref, Google Scholar
52 : Restraint use for psychiatric patients in the pediatric emergency department. Pediatr Emerg Care 2006; 22:7–12Crossref, Google Scholar
53 : Agitation treatment for pediatric emergency patients. J Am Acad Child Adolesc Psychiatry 2008; 47:132–138Crossref, Google Scholar
54 : Crisis in the emergency department: the evaluation and management of acute agitation in children and adolescents. Child Adolesc Psychiatr Clin N Am 2018; 27:367–386Crossref, Google Scholar
55 : Brief Rating of Aggression by Children and Adolescents (BRACHA): a reliability study. J Am Acad Psychiatry Law 2012; 40:374–382Google Scholar
56 : Using the Dynamic Appraisal of Situational Aggression with mental health inpatients: a feasibility study. Patient Prefer Adherence 2016; 10:691–701Crossref, Google Scholar
57 : The Dynamic Appraisal of Situational Aggression: an instrument to assess risk for imminent aggression in psychiatric inpatients. Behav Sci Law 2006; 24:799–813Crossref, Google Scholar
58 : The Overt Aggression Scale Modified (OAS-M) for clinical trials targeting impulsive aggression and intermittent explosive disorder: validity, reliability, and correlates. J Psychiatr Res 2020; 124:50–57Crossref, Google Scholar
59 : Aggressive behaviour among in-school adolescents in a developing country: patterns and associated factors. Int J Adolesc Med Health 2020; 34:171–177Crossref, Google Scholar
60 : Practice parameter for the assessment of the family. J Am Acad Child Adolesc Psychiatry 2007; 46:922–937Crossref, Google Scholar
61 : Practitioner review: clinical applications of attachment theory and research for infants and young children. J Child Psychol Psychiatry 2011; 52:819–833Crossref, Google Scholar
62 : An approach to interviewing adolescents. Paediatr Child Health 2003; 8:554–556Crossref, Google Scholar
63 : Management of agitation in individuals with autism spectrum disorders in the emergency department. Child Adolesc Psychiatr Clin N Am 2014; 23:83–95Crossref, Google Scholar
64 : Use of cognitive behavioral therapy and token economy to alleviate dysfunctional behavior in children with attention-deficit hyperactivity disorder. Front Psychiatry 2015; 6:167Crossref, Google Scholar
65 : Enteral guanfacine to treat severe anxiety and agitation complicating critical care after cardiac surgery. Semin Cardiothorac Vasc Anesth 2018; 22:403–406Crossref, Google Scholar
66 : Guanfacine extended release for the reduction of aggression, attention-deficit/hyperactivity disorder symptoms, and self-injurious behavior in Prader-Willi syndrome—a retrospective cohort study. J Child Adolesc Psychopharmacol 2019; 29:313–317Crossref, Google Scholar
67 : Managing disruptive behaviour in autism-spectrum disorder with guanfacine. J Psychiatry Neurosci 2018; 43:359–360Crossref, Google Scholar
68 : Recommendations for pharmacological management of inpatient aggression in children and adolescents. Psychiatry 2010; 7:32–40Google Scholar
69 : What do we really know about PRN use in agitated children with mental health conditions: a clinical review. Evid Based Ment Health 2018; 21:166–170Crossref, Google Scholar
70 : Adverse effects of psychotropic medications in children: predictive factors. J Can Acad Child Adolesc Psychiatry 2014; 23:218–225Google Scholar
71 : The neural effects of psychotropic medications in children and adolescents. Child Adolesc Psychiatr Clin N Am 2012; 21:753–771Crossref, Google Scholar
72 : Potential drug-drug interactions with psychotropic drugs in paediatric inpatients: a cross-sectional study. Int J Clin Pract 2021; 75:e14107Crossref, Google Scholar
73 : Treatment recommendations for the use of antipsychotics for aggressive youth (TRAAY): part II. J Am Acad Child Adolesc Psychiatry 2003; 42:145–161Crossref, Google Scholar
74 : Treatment recommendations for the use of antipsychotics for aggressive youth (TRAAY): part I. a review. J Am Acad Child Adolesc Psychiatry 2003; 42:132–144Crossref, Google Scholar
75 : Dosing and monitoring: children and adolescents. Psychopharmacol Bull 2018; 48:34–92Google Scholar
76 : The art and science of drug titration. Ther Adv Drug Saf 2020; 11:2042098620958910Crossref, Google Scholar
77 : Medication dosage in overweight and obese children. J Pediatr Pharmacol Ther 2017; 22:81–83Google Scholar
78 : Canadian Network for Mood and Anxiety Treatments (CANMAT) 2016 clinical guidelines for the management of adults with major depressive disorder: section 3. pharmacological treatments. Can J Psychiatry 2016; 61:540–560Crossref, Google Scholar
79 : Depression in medically ill children and adolescents. Child Adolesc Psychiatr Clin N Am 2019; 28:421–445Crossref, Google Scholar



